MitoBright LT Deep Red
Mitochondrial Staining
- High Intracellular Retention
- Long-Term Visualization of Mitochondria
- Allows for Collagen-coated Plates
-
Product codeMT12 MitoBright LT Deep Red
| Unit size | Price | Item Code |
|---|---|---|
| 400 μl x 1 | $169.00 | MT12-12 |
Description
Product Description
A mitochondrion is a multifunctional organelle not only involved in energy production in a cell, but other additional cellular functions. The active cycle of mitochondrial fusion and division induces morphological changes, which is called mitochondrial dynamics. Abnormalities in morphological control of mitochondria are associated with neurodegenerative diseases, metabolic disorders, aging, and so on. Therefore, the demand for long-term observation of mitochondrial dynamics has recently been increasing.
Methods for monitoring mitochondrial morphology, dynamics, and number are usually based on small fluorescent molecules or plasmid transfection techniques. The use of plasmids requires the target protein to be stably expressed, while small fluorescent molecules are widely used because they can simply be added to cells. Among commercially available small fluorescent molecules, those containing the chloromethyl moiety are commonly used. However, these dyes have some limitations, including short-term retention in cells, decreased fluorescence intensity in serum, and high background.
Dojindo’s MitoBright LT dyes overcome these limitations. MitoBright LT dyes are designed to exhibit mitochondria retention for long-term visualization. In addition, the MitoBright LT dyes show stronger fluorescence signals compared with other commercially available dyes that contain the chloromethyl moiety. The MitoBright LT dyes offer three different color options (Green, Red and Deep Red), and are provided as a ready-to-use DMSO solution. A working solution can easily be prepared in a single dilution step with growth medium or HBSS.
Manual
Technical info
HeLa cells were washed with HBSS (Hank’s Balanced Salt Solution) and subsequently stained with each of MitoBright LTs or an existing reagent. The culture medium was replaced with serum-containing medium, and mitochondria were observed after 4 days of incubation. As a result, fluorescence intensity of an existing reagent decreased significantly after 4 days, but in MitoBright LT, fluorescence intensity remained unchanged and mitochondria were clearly observable. Moreover, after further incubation, we confirmed that MitoBright LT was retained in mitochondria even after 7 days.
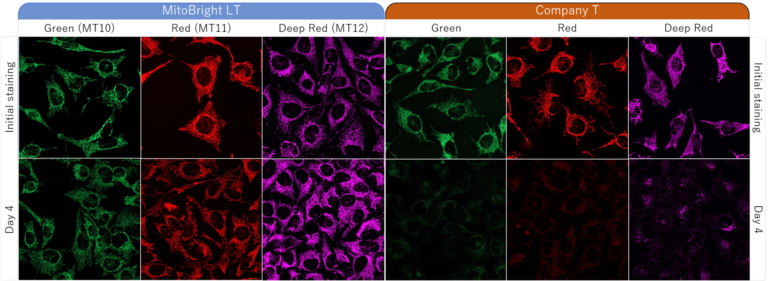
Dojindo Laboratories' Long-term mitochondrial staining reagent MitoBright LT series is widely used around the world.
| Product Name | Samples | Observation time after staining |
Instrument | Publications |
|---|---|---|---|---|
| MitoBright LT Green |
Cell(C2C12) |
5 Days | Fluorescence microscope | Polymers |
| MitoBright LT Green | Cell(BMM) | 36 hrs or 3 Days | Fluorescence microscope/ Flow Cytometer |
JCI Insights |
| MitoBright LT Red | Cell(A11, P29) | 3 Days | Fluorescence microscope | BMC Mol. Cell Biol. |
| MitoBright LT Deep Red | Cell(HT-1080, MCF-10A, MCF-7 ) | 8 hrs | Fluorescence microscope | Advanced Therapeutics |
| MitoBright LT Deep Red | Cell(4T1) | 24 hrs | Fluorescence microscope | Advanced Functional Materials |
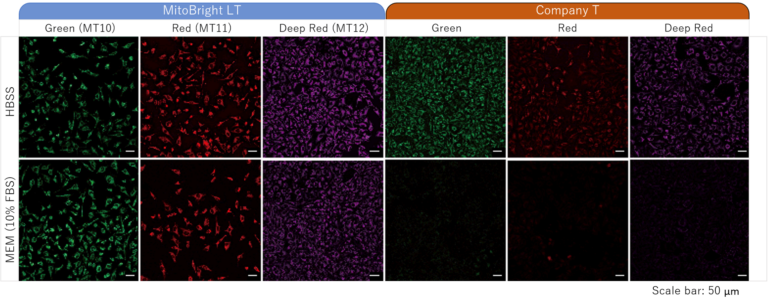
Stained in serum-contained media
Staining was performed with either MitoBright LT or an existing reagent in serum-containing or serum-free medium. In serum-containing medium, low fluorescence intensity was observed when using the existing reagent, however the cells stained with MitoBright LT experienced clear, resilient fluorescence of mitochondria
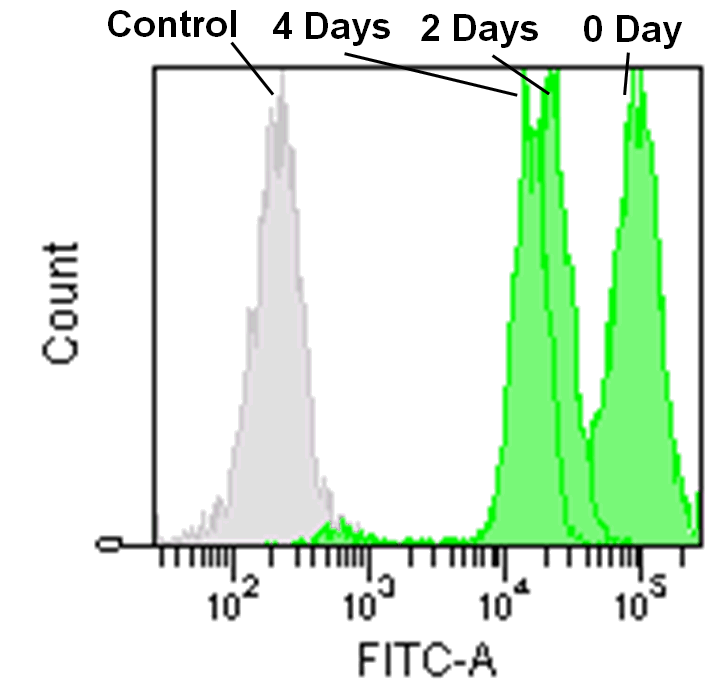
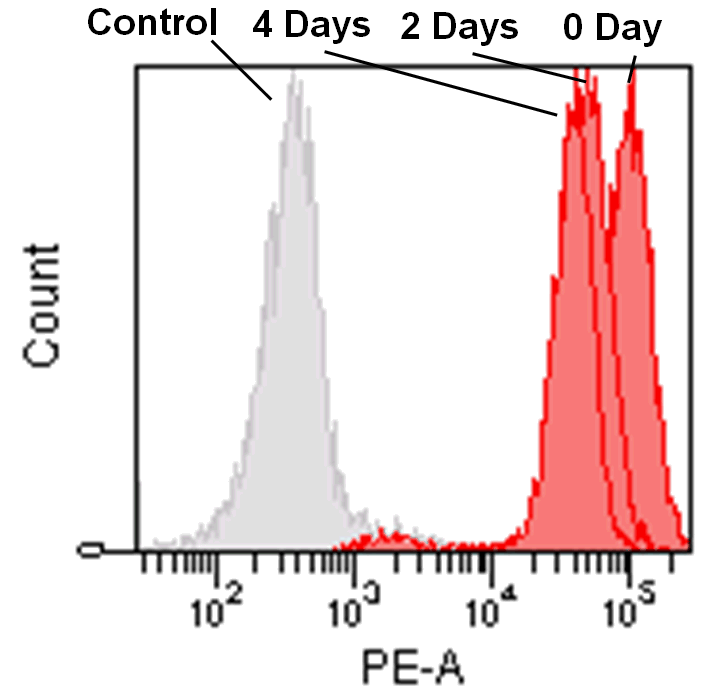
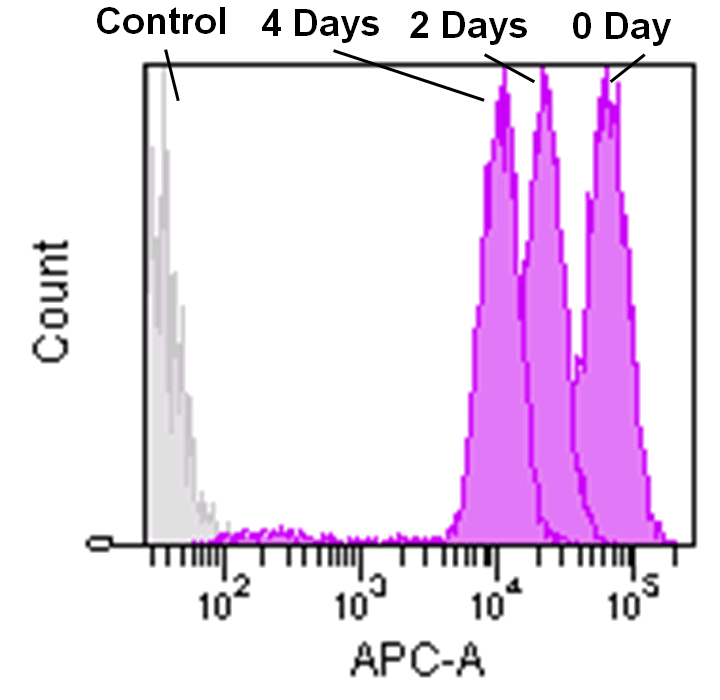
Detection by flow cytometry
Jurkat cells (3.2×105 cells/mL) were suspended in RPMI medium supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin, plated on a 35cm dish, and incubated overnight at 37˚C in a 5% CO2 atmosphere. RPMI medium was removed and replaced with 5mL of MitoBright LT working solution (0.1μmol/L). The cells were then incubated for 30 minutes at 37˚C. The working solution was removed and the cells were washed twice with 5mL of RPMI medium. Fresh RPMI medium was poured into the dish and cells were passaged and analyzed every 2 days by flow cytometry.
Mitochondrial membrane potential dependence of MitoBright LT Deep Red
Mitochondrial condition in the FCCP treated HeLa cells was compared with untreated cells using MitoBright LT Deep Red or existing reagents by flow cytometry.
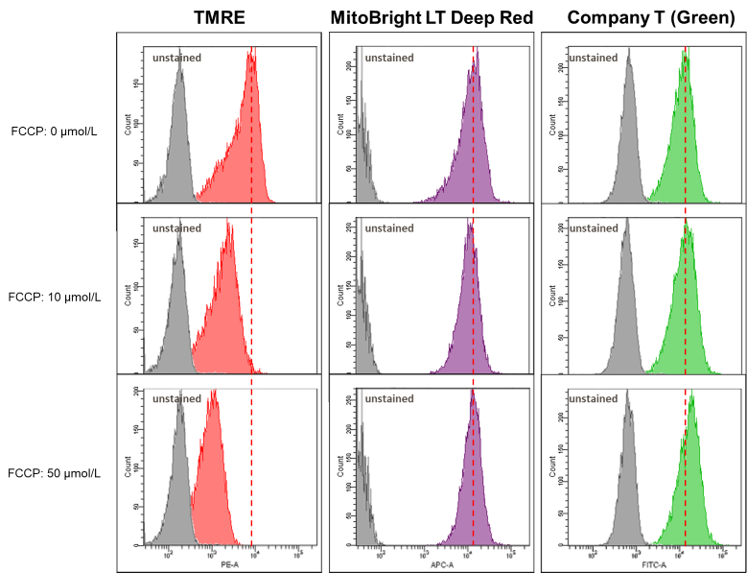
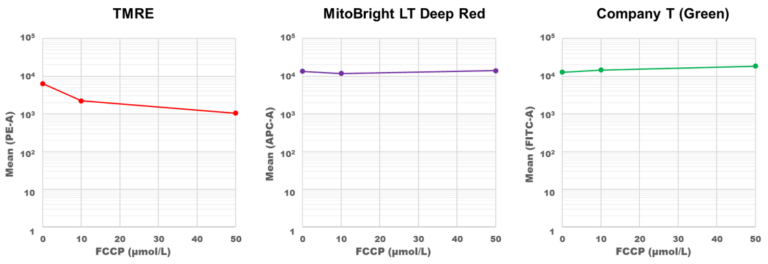
Result:
The fluorescence intensity of TMRE decreased significantly when treated with FCCP, but in MitoBright LT Deep Red and an existing reagent (Company T), fluorescence intensity remained unchanged. MitoBright LT Deep Red retention may not depend on membrane potential.
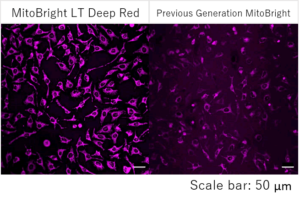
Detection of mitochondria using a collagen coated tissue plate
Collagen-coated tissue culture plates are often used for examination of mitochondrial morphology because this imaging requires high magnifications.
The existing mitochondrial staining reagent bound to collagen, resulting with an increase in background staining, whereas the MitoBright LT series yielded clearly stained mitochondria.
Conditions:
Staining: HeLa cells were plated on a collagen coated tissue culture plate and cultured for 24 hours. The culture medium was removed, and the cells were washed with HBSS.
MitoBright LT Deep Red working solution (100 nmol/L) was poured into the plate, the cells were incubated for 30 minutes. The solution was removed, and the cells were washed with HBSS. A fluorescence microscope was used for imaging of mitochondria.
Detection: Ex: 640 nm, Em: 650-700 nm
Result:
In mitochondria stained with an existing reagent, background staining was observed, but the mitochondria stained with MitoBright LT were clearly stained without having effect of background staining.
Fixed cell imaging
1. HeLa cells were seeded on a μ-slide 8 well plate and cultured at 37°C overnight in a 5% CO2 incubator.
2. The supernatant was removed and the cells were washed with serum-containing medium twice.
3. 0.1 μmol/l MitoBright LT (Green/Red/Deep Red) working solution (serum-containing medium) was added and the cells were incubated at 37°C for 15-60 min in the 5% CO incubator.
4. The supernatant was removed and the cells were washed with PBS twice.
5. 4% paraformaldehyde (PFA) solution was added and the cells were fixed at room temperature for 15 minutes.
6. The supernatant was removed and the cells were washed with PBS twice.
7. The cells were observed under a fluorescence microscope.
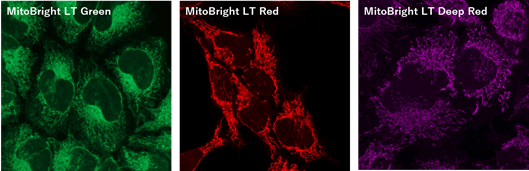
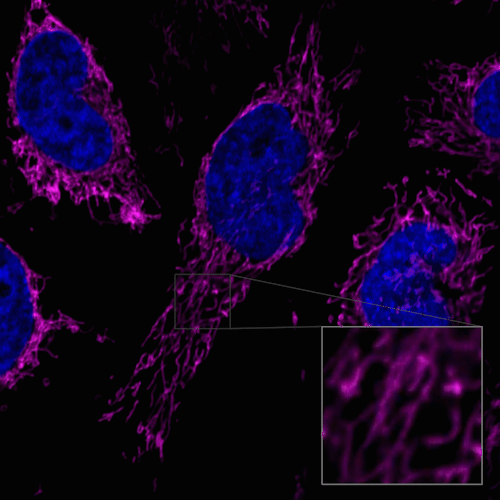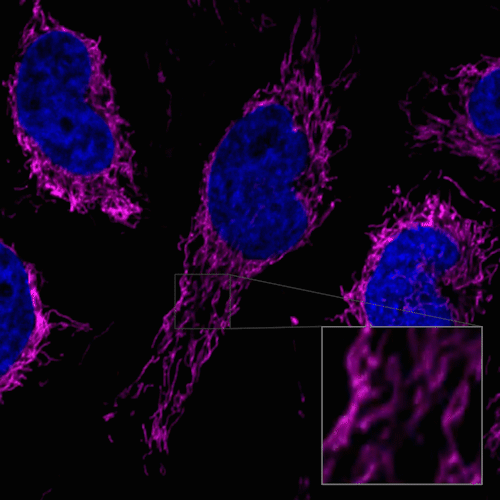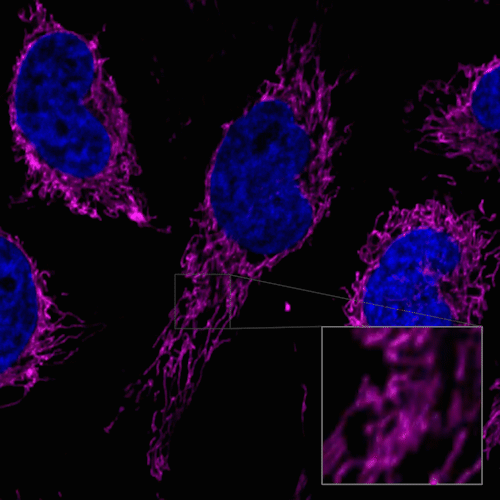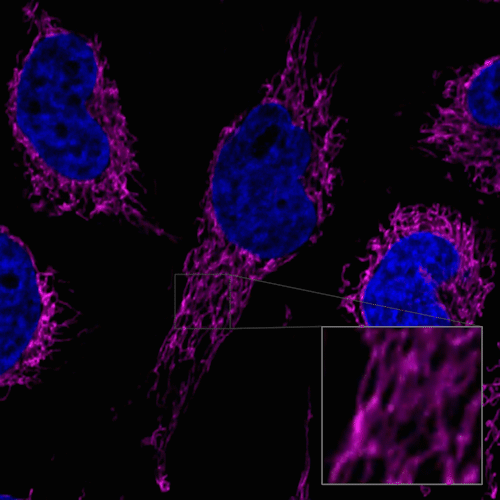
Figure 1. Fluorescent images of 4% PFA fixed HeLa cells (Stain then Fix)
Note: Cells should be fixed with paraformaldehyde (PFA). Fixation with methanol or other solvents extracts lipids and results in poor staining.
Note: The staining has low tolerance for permeabilization after fixation, and cannot be used with detergent.
(Triton X-100, NP-40 etc.)
Product Comparison
Observation of the internal structure of mitochondria using a super-resolution laser microscope (STED)
Cytoplasmic hybrid (Cybrid) cells with the causative mutation of mitochondrial disease were stained with MitoBright LT Deep Red and observed using a super-resolution laser microscope, which revealed abnormal mitochondrial cristae structure.
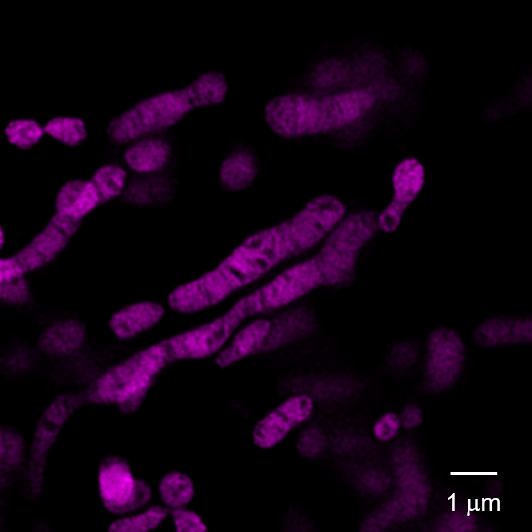
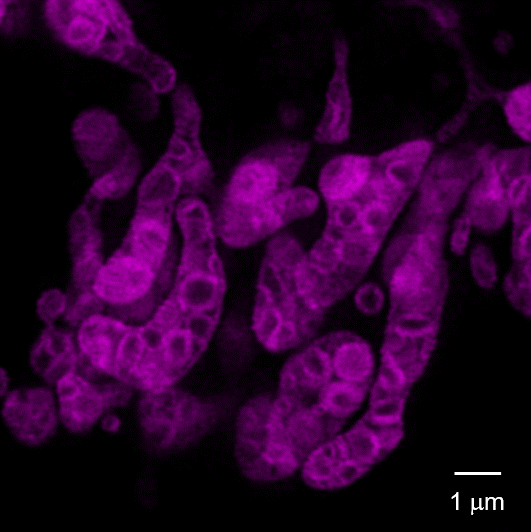
Left panel: Control Cybrid cells, Right Panel: Cybrid cells with the causative mutation of mitochondrial disease
Dye: MitoBright LT Deep Red (100 nmol/l)
Equipment: Leica Super Resolution Laser Microscope TCS SP8 STED 3X
Ex. 640 nm / Em. 650-700 nm
STED laser: 775 nm
<Experimental Procedure>
1. Seed cells in a glass base dish and incubate in an incubator (37°C, 5% CO2) for 2 days.
2. Remove the culture medium and add MitoBright LT Deep Red (100 nmol/l) prepared in L-15 medium (containing 10% FBS).
3. Incubate in an incubator (37°C, 5% CO2) for 45 minutes.
4. Remove supernatant and wash twice with HBSS.
5. Add L-15 medium (containing 10% FBS) and observe with a super-resolution laser microscope (Leica TCS SP8 STED 3X).
This data was kindly provided by Dr. Ikuro Osawa and Dr. Yasunori Fujita, Research Team for Regulation of Aging, Tokyo Metropolitan Institute for Geriatrics and Gerontology.
Observation of mitochondrial fission and fusion
HeLa cells were stained with MitoBright LT Deep Red, then replaced with medium-containing serum and observed for 30 minutes for mitochondrial morphology.
<Measuring equipment>
Confocal laser microscope, magnification 63x
<Observation conditions>
Ex 640 nm, Em 650-700 nm
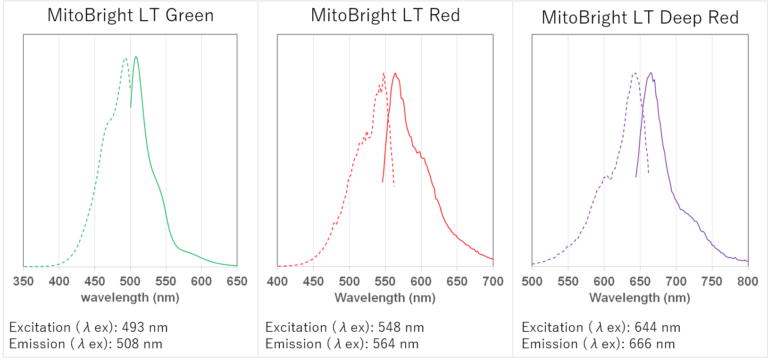
Fluorescence Properties
References
| No. | Sample | Instrument | Reference(Line) |
|---|---|---|---|
| 1 | Cell (HT-1080; MCF-10A; MCF-7; HCT-116 ) | Microscope | L. Zhou, Q. Guan, W. Li, Z. Zhang, Y. Li and Y. Dong, "A Ferrocene-Functionalized Covalent Organic Framework for Enhancing Chemodynamic Therapy via Redox Dyshomeostasis", 2021, doi:10.1002/smll.202101368. |
| 2 | Cell (HT-1080; MCF-10A; MCF-7 ) | Microscope | Q. Guan, L. Zhou, Y. Dong, "Construction of Nanoscale Covalent Organic Frameworks via Photocatalysis-Involved Cascade Reactions for Tumor-Selective Treatment", 2021, doi:10.1002/adtp.202100177. |
| 3 | Cell (4T1) | Microscope | X. Chen, X Yin, L. Zhan, J. Zhang, Y. Zhang, Y. Wu, J. Ju, Y. Li, Q. Xue, X. Wang, C. Li, R. R. L. Reis and Y. Wang, "Organelle-Specific Anchored Delivery System Stretching a Reversal of Tumor Hypoxia Microenvironment to a Combinational Chemo-Photothermal Therapy", 2021, doi:10.1002/adfm.202108603. |
| 4 | Cell (SW982) | Microscope | S. Xia, Z. Ma, B. Wang, F. Gao, C. Yi, X. Zhou, S. Guo, L. Zhou, "In vitro anti-synovial sarcoma effect of diallyl trisulfide and mRNA profiling", 2021, doi:10.1016/j.gene.2021.146172. |
| 5 | AIG-1 | Microscope | Y. Fujita, M. Iketani, M. Ito, I. Ohsawa, "Temporal changes in mitochondrial function and reactive oxygen species generation during the development of replicative senescence in human fibroblasts", 2022, doi:10.1016/j.exger.2022.111866. |
Q & A
-
Q
Does MitoBright LT Deep Red in DMSO solution not degrade after repeated freeze-thaw cycles?
-
A
We have confirmed that staining was possible using a solution that was subjected to 30 cycles of freeze-thaw cycles.
-
Q
Does depolarization after MitoBright LT staining affect fluorescent intensity?
-
A
Effects are confirmed by depolarization conditions and cell type. Also, the degree of influence differs for each MitoBright LT dye.
For reference, we stained HeLa cells with each MitoBright LT dye and observed changes in fluorescent staining after depolarization under the following conditions.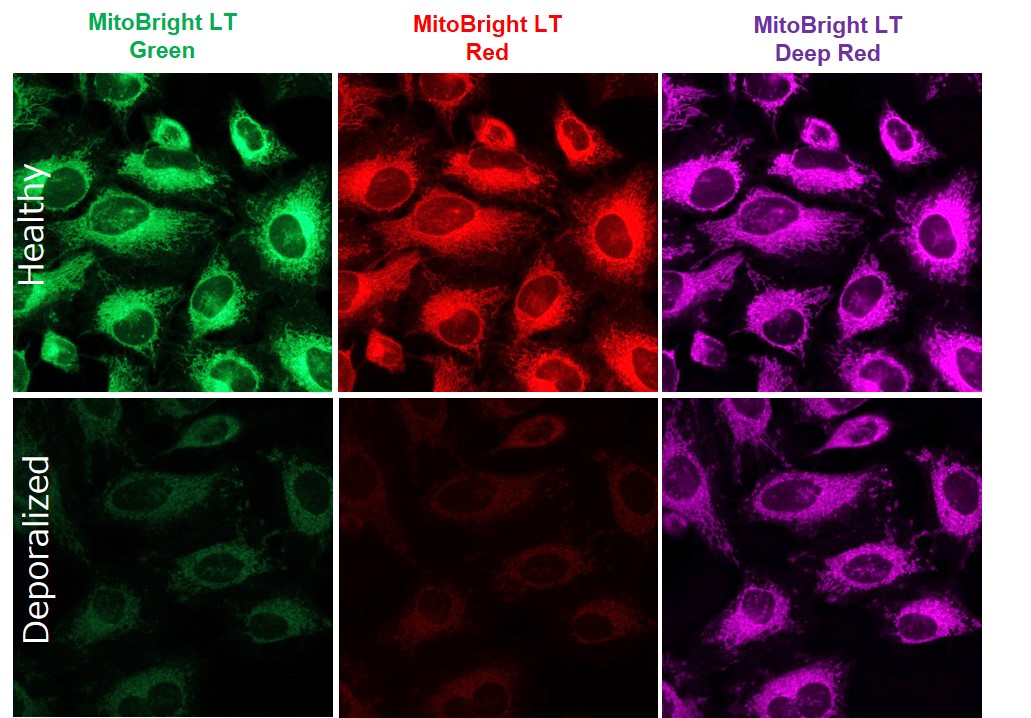
<Staining conditions>
HeLa cells were stained with MitoBright LT dyes (100 μmol/l, 30 min incubation), washed with HBSS, treated with FCCP (100 μmol/l, 60 min incubation), washed twice with HBSS, and observed.<Detection conditions>
MitoBright LT Green: Ex 488 nm/Em 500-560 nm
MitoBright LT Red: Ex 561 nm/Em 560-620 nm
MitoBright LT Deep Red: Ex 640 nm/Em 650-700 nm
Handling and storage condition
| Appearance: | Blue liquid |
|---|---|
| Absorbance: | 0.300~0.500 |
| -20°C, Protect from light | |
|
Danger / harmful symbol mark |

|
|---|---|