MT-1 MitoMP Detection Kit
Mitochondrial Membrane Potential Detection
- Applicable to live cells and fixation after staining
- Monitoring of mitochondrial membrane potential
- Highly sensitive detection of changes in mitochondrial membrane potential
-
Product codeMT13 MT-1 MitoMP Detection Kit
| Unit size | Price | Item Code |
|---|---|---|
| 1 set | Find your distributors | MT13-10 |
The general number of usable assays per 1 set 35 mm dish x 30
| 1 set | MT-1 Dye Imaging Buffer (10x) |
20 μl x 3 11 ml x 1 |
|---|
Description
Mitochondria is an important organelle that uses oxygen to synthesize ATP, producing the necessary energy for living cells to thrive1). Decreased mitochondrial activity and mitochondrial dysfunction are associated with cancer, aging, and neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease2,3). Therefore, mitochondrial membrane potential (MMP) has been widely studied as a promising target for mitochondria-related diseases.
Reference
1) K. F. Ferri, et al., J. Exp. Med., 2000, 192, 1081.
2) N. Matsuda, et al., J. Cell Biol., 2010, 189, 211.
3) J. L. Wang, et al., PNAS, 2000, 97, 7124.
Manual
Technical info
Comparison of reagents for mitochondrial membrane potential detection
| Features | Sensitivity | Fixation | Monitoring | Fluorescence change (upon loss of mitochondrial membrane potential) |
Detection |
|
|---|---|---|---|---|---|---|
|
Recomended for starting-up |
✓ |
|
|
Color change from red to green |
|
|
|
MT-1 |
Recommended for more detailed analysis |
✓ (High) |
✓ |
✓ |
Decrease in fluorescence intensity |
530-560 nm / 570-640 nm |
|
TMRE |
Widely used |
✓ (High) |
|
|
Decrease in fluorescence intensity |
530-560 nm / 570-640 nm |
Overcome three limitations of conventional reagents
JC-1 dye, TMRE, and TMRM are widely used to monitor MMP, however, these dyes have some limitations, such as low photostability and poor retention after aldehyde fixation. These limitations result in poor reproducibility of experiments.
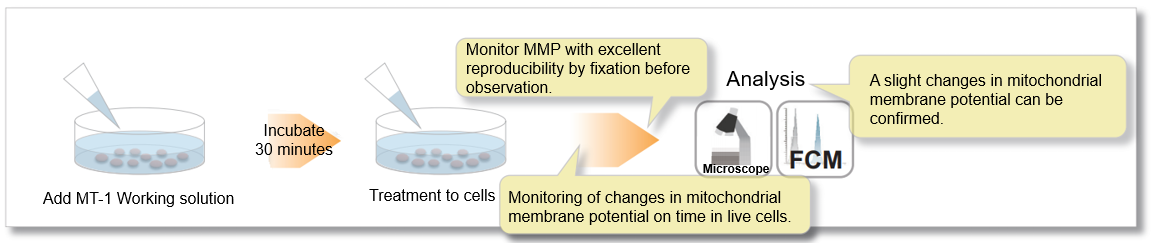
Dojindo’s MT-1 MitoMP Detection Kit overcomes these limitations. In addition, the Imaging Buffer included in this kit minimizes background fluorescence and maintains cell vitality while the assay is being performed.
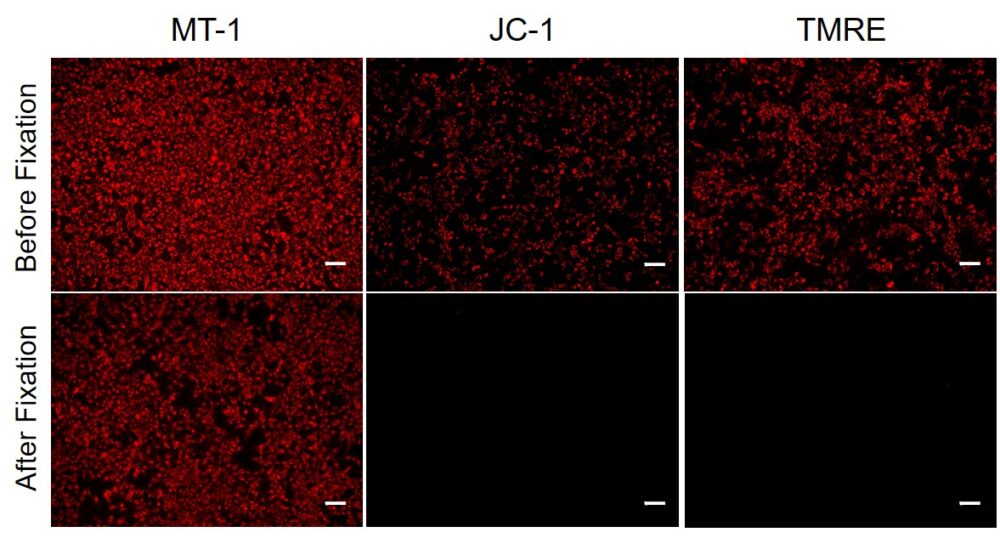
① Applicable to fixation after staining
Since mitochondrial membrane potential (MMP) fluctuates according to slight changes of cellular status, extreme care has been needed to obtain repeatable data. In JC-1 dye, which is widely used for measuring the MMP, the fluorescence is lost after a cell has been fixed; therefore, the use of living cells is necessary for the quick measurement of MMP.
In the meantime, the MT-1 dye, which remains unquenched even in the cells fixed with paraformaldehyde after staining, can be used to conduct a highly repeatable experiment.
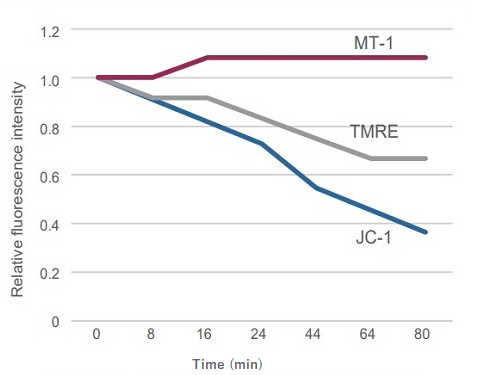
② Allow to monitor mitochondrial membrane potential
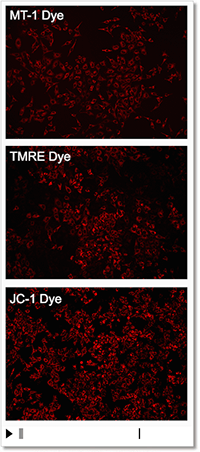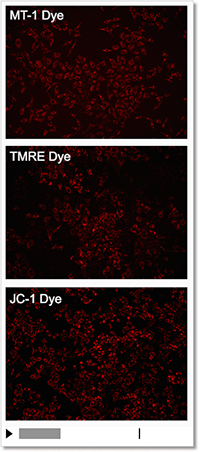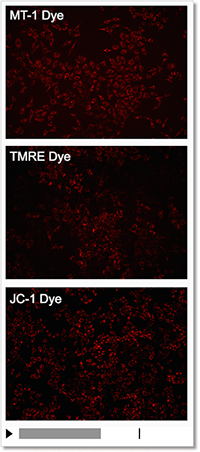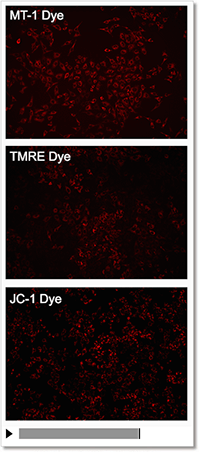
Detection Condition
Ex: 530-560 nm, Em: 570-640 nm
Scale bar: 100 μm
HeLa cells
③ High sensitivity for mitochondrial membrane potential
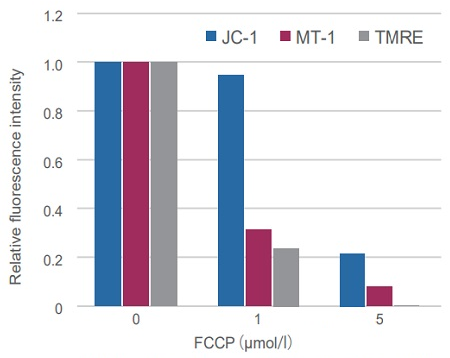
Sometimes it is difficult to detect slight changes of MMP in JC-1 stained mitochondria. In such a case, tetramethylrhodamine ethyl ester (TMRE) was used to monitor MMP. MT-1 can provide equivalent detection sensitivity to TMRE.
Procedure
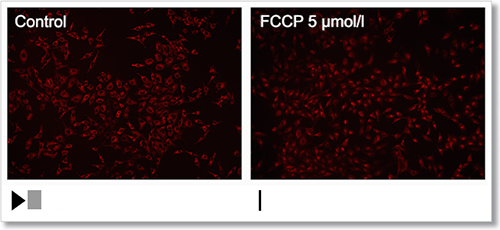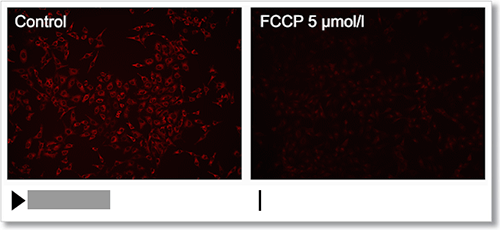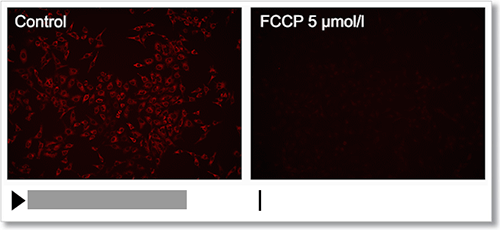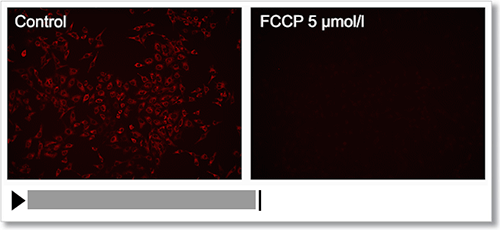
Experimental Example: Depolarization
HeLa cells were treated with a depolarizing agent, carbonyl cyanide-p-trifluoromethoxyphenylhydrazone (FCCP), and changes in mitochondrial membrane potential were observed in a time-lapse imaging with this kit.
As a result, it was confirmed that the mitochondrial membrane potential of the cells treated with FCCP decreased.
Experimental Example: Changes in mitochondrial membrane potential in apoptosis-induced cells
Apoptosis was induced by Etoposide to HL60 cells stained with MT-1 in advance, then co-stained with Annexin V-FITC and analyzed by flow cytometry.
Apoptotic progress and MMP change were confirmed through the fluorescence intensity changes of Annexin V-FITC (an increase in the green fluorescence intensity) and MT-1 (a decrease in the red fluorescence intensity), respectively.
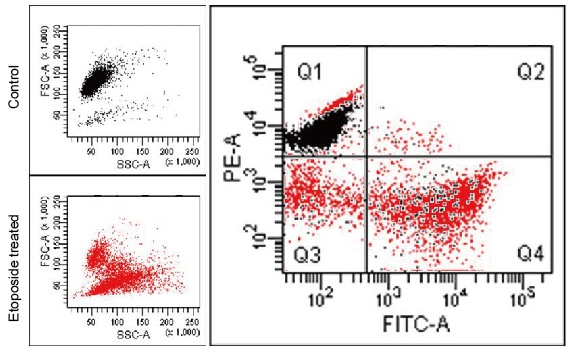
Detection Condition
Annexin V-FITC (green): Ex. 488 nm / Em. 500 – 560 nm
Mitochondria membrane potential (MT-1, red): Ex 488 nm / Em 564-604 nm
Application Data: Simultaneously evaluation of mitochondrial superoxide and membrane potential
After HeLa cells were washed with HBSS, co-stained with MT-1 MitoMP Detection Kit and mitochondrial superoxide detection dye (MitoBright ROS Deep Red: Code MT16), and the generated mitochondrial ROS and membrane potential were observed simultaneously. As a result, the decrease in mitochondrial membrane potential and the generation of mitochondrial ROS are simultaneously observed.
<General Protocol >
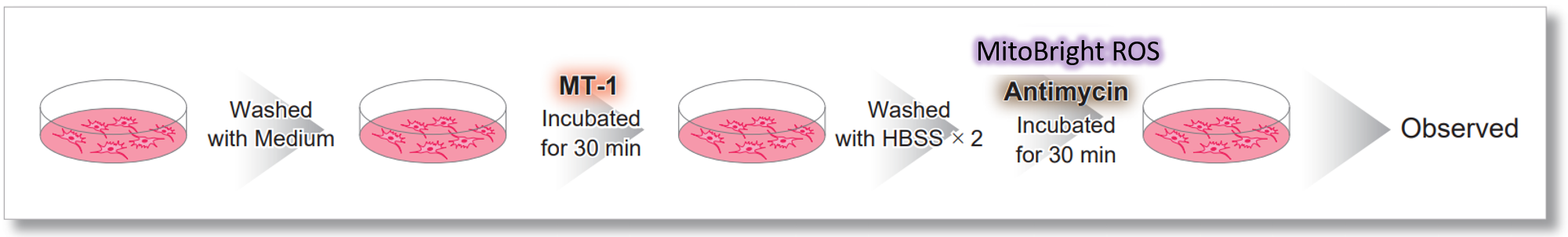

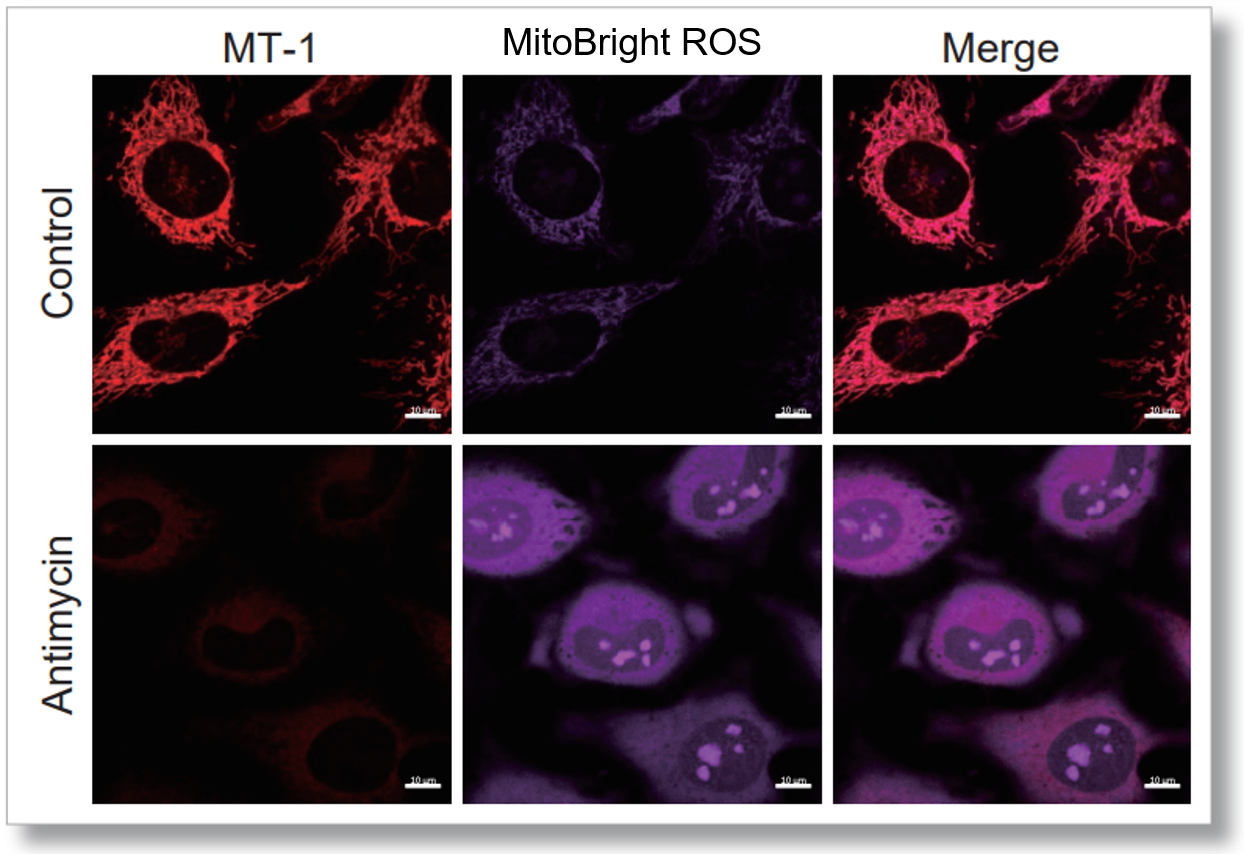
<Imaging Conditions>(Confocal microscope)
MT-1: Ex=561, Em=560-600 nm
MitoBright ROS: Ex=633 nm, Em=640-700 nm
Scale bar: 10 μm
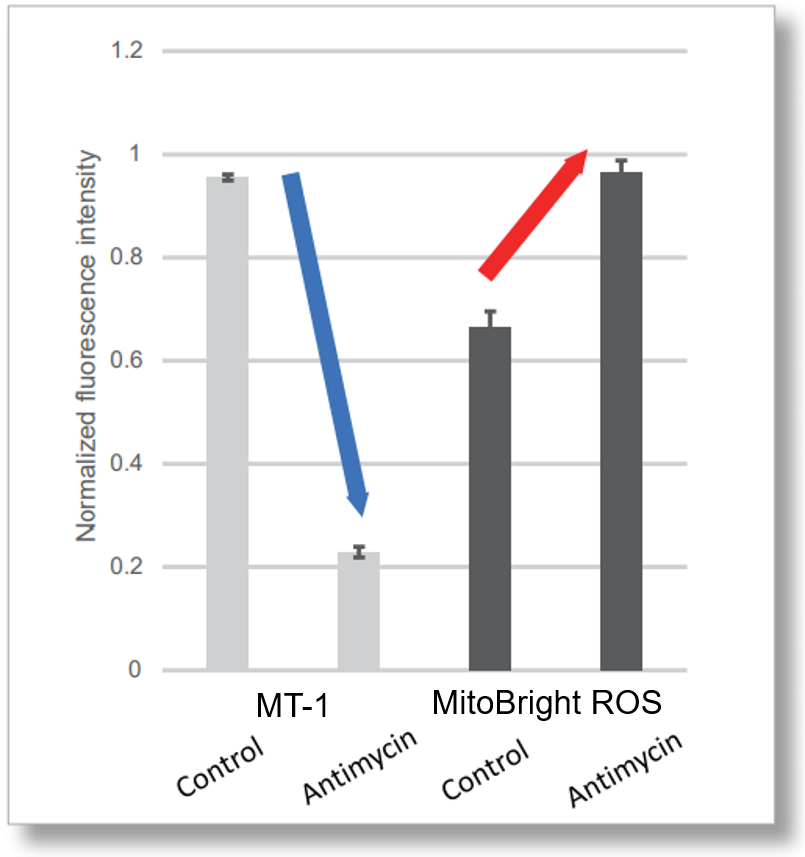


<Examination Conditions>(Plate Reader)Tecan, Infinite M200 Pro
MT-1: Ex=540-550 nm, Em=590-610 nm (Gain=200)
MitoBright ROS: Ex=545-555 nm, Em = 665-685 nm
References
| No. | Sample | Instrument | Reference |
|---|---|---|---|
| 1 | STHdh Cells | Microscope | N. Okada, T. Yako, S. Nakamura, M. Shimazawa, H. Hara, "Reduced mitochondrial complex II activity enhances cell death via intracellular reactive oxygen species in STHdhQ111 striatal neurons Q1 with mutant huntingtin", J. Pharmacol. Sci., 2021, doi:10.1016/j.jphs.2021.09.001. |
| 2 | Panc-1 Cells | Microscope | N. Okuni, Y. Honma, T. Urano, K. Tamura, "Romidepsin and tamoxifen cooperatively induce senescence of pancreatic cancer cells through downregulation of FOXM1 expression and induction of reactive oxygen species/lipid peroxidation", Mol. Biol. Rep., 2022, doi:10.1007/s11033-022-07192-9. |
| 3 | BM Cells | Microscope | Y. Aoyagi, Y. Hayashi, Y. Harada, K. Choi, N. Matsumura, D. Sadato, Y. Maemoto, A. Ito, S. Yanagi, D. Starczynowski, H. Harada, "Mitochondrial Fragmentation Triggers Ineffective Hematopoiesis in Myelodysplastic Syndromes", Cancer Discovery, 2022, doi:10.1158/2159-8290.CD-21-0032. |
| 4 | Flies indirect flight muscle Cells | Microscope | N. Nozawa, M. Noguchi, K. Shinno, M. Tajima, S. Aizawa, T. Saito, A. Asada, T. Ishii, M. Ishizuka, K. Iijima and K. Ando, "5-Aminolevulinic acid and sodium ferrous citrate ameliorate muscle aging and extend healthspan in Drosophila", FEBS Open Bio, 2022, doi:10.1002/2211-5463.13338. |
| 5 | HBME Cells | Microscope | Y. Sakai, M. Taguchi, Y. Morikawa, H. Miyazono, K. Suenami, Y. Ochiai, E. Yanase, T. Takayama, A. Ikari, T. Matsunaga, "Apoptotic mechanism in human brain microvascular endothelial cells triggered by 40-iodo-α-pyrrolidinononanophenone: Contribution of decrease in antioxidant properties", Toxicol. Lett., 2022, doi:10.1016/j.toxlet.2021.11.018. |
| 6 | MIN6-M9 Cells | Microscope | R. Inoe, T. Tsuno, Y. Togashi, T. Okuyama, A. Sato, K. Nishiyama, M. Kyohara, J. Li, S. Fukushima, T. Kin, D. Miyashita, Y. Shiba, Y. Atobe, H. Kiyonari, K. Bando, A. S. Shapiro, K. Funakoshi, R. N. Kulkarni, Y. Terauchi, and J. Shirakawa, "Uncoupling protein 2 and aldolase B impact insulin release by modulating mitochondrial function and Ca2+ release from the ER", 2022, iScience, doi:10.1016/j.isci.2022.104603. |
| 7 | SH-SY5Y Cells | Flow Cytometer | M. Hashimoto, M. Fujimoto, K. Konno, M. L. Lee, Y. Yamada, K. Yamashita, C. Toda, M. Tomura, M. Watanabe, O. Inanami and H. Kitamura, "Ubiquitin-Specific Protease 2 in the Ventromedial Hypothalamus Modifies Blood Glucose Levels by Controlling Sympathetic Nervous Activation", J. Neurosci., 2022, doi:10.1523/JNEUROSCI.2504-21.2022. |
| 8 | Fibroblasts, ciBAs | Microscope | Y. Takeda and P. Dai, "Chronic Fatty Acid Depletion Induces Uncoupling Protein 1 (UCP1) Expression to Coordinate Mitochondrial Inducible Proton Leak in a Human-Brown-Adipocyte Model", 2022, doi:10.3390/cells11132038. |
| 9 | Sperm cells from C. osakensis queens | Microscope | A. Gotoh, M. Takeshima and K Mizutani, "Near-anoxia induces immobilization and sustains viability of sperm stored in ant queens", Sci. Rep., 2023, doi:10.1038/s41598-023-29705-7. |
| 10 | Nucleus Pulposus Cells | Microscope | K. Suyama, D. Sakai, S. Hayashi, N. Qu, H. Terayama, D. Kiyoshima, K. Nagahori and M. Watanabe, "Bag-1 Protects Nucleus Pulposus Cells from Oxidative Stress by Interacting with HSP70", Biomedicines, 2023, doi:10.3390/biomedicines11030863. |
| 11 | HL60 Cells, KG1a Cells | Flow Cytometer | K. Kamachi, H. Ureshino, T. Watanabe, N. Y. Sakai, Y. F. Kurahashi, K. Kawasoe, T. Hoshiko, Y. Yamamoto, Y. Kurahashi, and S. Kimura , "Combination of a New Oral Demethylating Agent, OR2100, and Venetoclax for Treatment of Acute Myeloid Leukemia", Cancer Res Commun., 2023, doi:10.1158/2767-9764.CRC-22-0259. |
| 12 | RAW264 Cells | Microscope | H. Gu, Y. Zhu, J. Yang, R. Jiang, Y. Deng, A. Li, Y. Fang, Q. Wu, H. Tu, H. Chang, J. Wen and X. Jiang, "Liver-Inspired Polyetherketoneketone Scaffolds Simulate Regenerative Signals and Mobilize Anti-Inflammatory Reserves to Reprogram Macrophage Metabolism for Boosted Osteoporotic Osseointegration", Adv sci, 2023, doi:10.1002/advs.202302136. |
Q & A
-
Q
Are there any tips for successful time-lapse imaging?
-
A
Please minimize the excitation light and increase the detection sensitivity.
Intermittent exposure of cells to excitation light may cause cell damage and degradation of the fluorescent dye, please optimize the interval time.
-
Q
Is it possible to fixed cells after staining with MT-1?
-
A
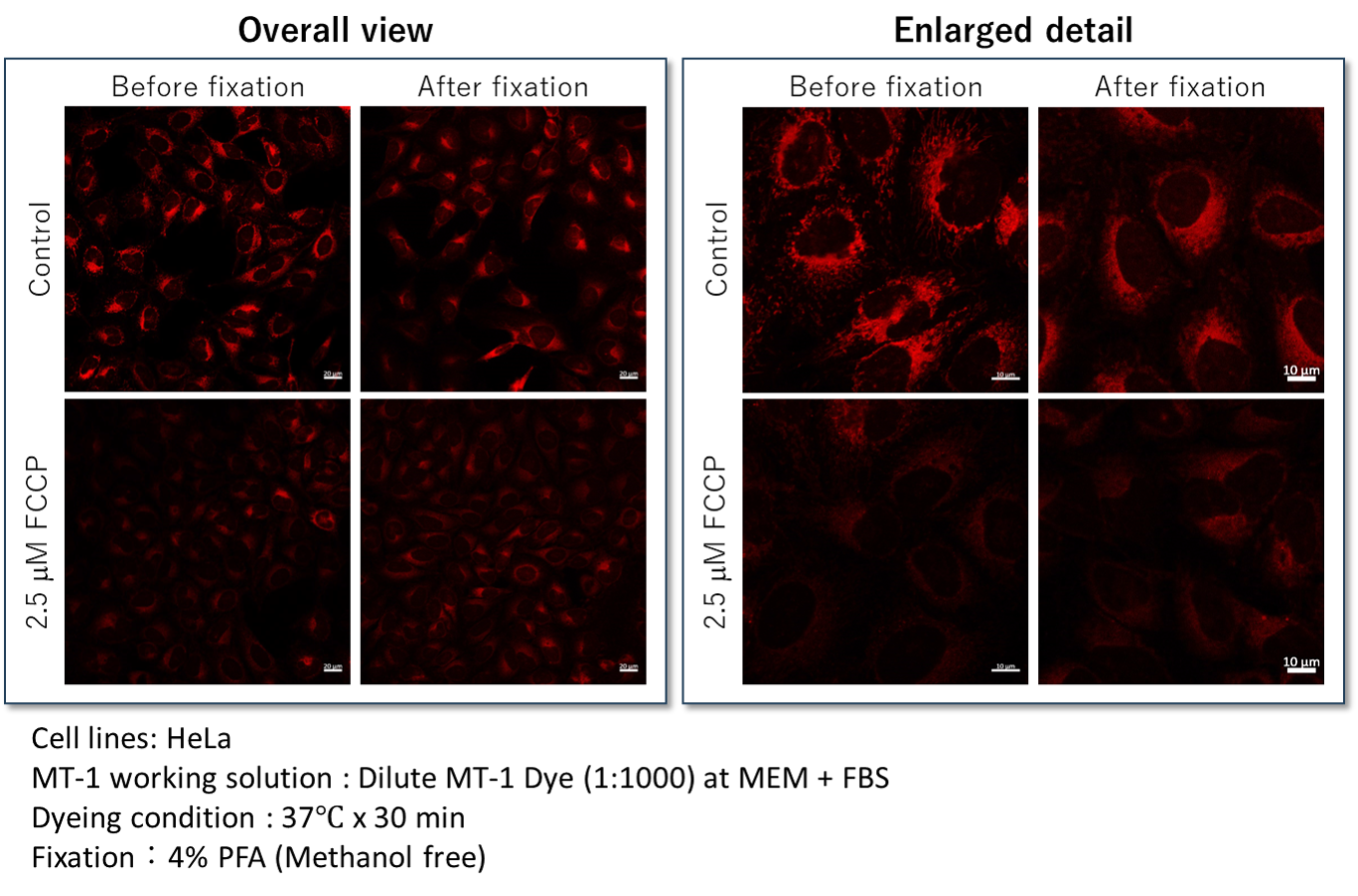
Yes, cells should be fixed with 4% paraformaldehyde (PFA), and cannot be used with detergents (Triton X-100, NP-40 etc.) because this step may induce leakage of the dye from the cells. (Recommended fixation time:15 - 30 minutes)
The fluorescence intensities should remain after fixation (left image, Overall view). In contrast to the fluorescence intensities, the localization of MT-1 Dye was different before and after fixation (right, Enlarged detail).
We recommend observing live cells if the sample should be visualized both mitochondrial membrane potential and structural information.
-
Q
Is it possible to stain cells after fixation?
-
A
Since MT-1 accumulates in mitochondria depending on the mitochondrial membrane potential, it is not applicable for staining after fixation.
-
Q
Are there any experimental example as a positive control when comparing fluorescence intensities?
-
A
As a positive control, an example of an experiment using FCCP (carbonylcyanide-p-trifluoromethoxyphenylhydrazone) can be found in the Technical Manual.
-
Q
What concentration of MT-1 Dye should be used when optimize staining conditions?
-
A
The concentration of MT-1 Dye is recommended to be diluted 1000 times. but please refer to the following when optimizing the staining conditions.
<Fluorescence intensity is weak>
Please optimize the following concentration: between 500-1,000 times dilution.<Non-specific adsorption is observed>
Please optimize the following concentration: between 1,000 and 2,000 times dilution.
-
Q
Can I use buffer for the preparation of MT-1 working solution?
-
A
Hanks’ HEPES and HBSS can be used. It can also be prepared using MEM, RPMI and MEM with 10 % FBS.
-
Q
Is it possible to observe after the addition of MT-1 working solution without washing step?
-
A
After staining, the samples can be observed without washing step.
However, we don’t recommend observing them for long term visualization without washing step because of the possibility of cytotoxicity.
We recommend to remove the supernatant and replace it with the medium.
-
Q
Is it possible to use PBS in place of HBSS for washing after staining with MT-1?
-
A
We recommend the use of HBSS to reduce the cell damage.
If you do not have HBSS, we recommend washing the cells with medium.
Handling and storage condition
| -20°C, Protect from light | |
|
Danger / harmful symbol mark |

|
|---|---|