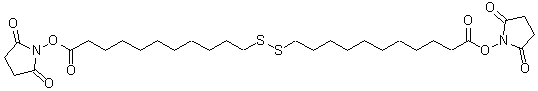Dithiobis(succinimidyl hexanoate)
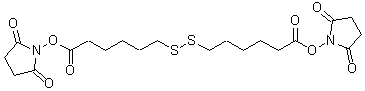
Cross-Linker
-
Product codeD539 Dithiobis(succinimidyl hexanoate)
-
CAS No.1083285-37-3
-
Chemical nameDithiobis(succinimidyl hexanoate)
-
MWC20H28N2O8S2=488.58
| Unit size | Price | Item Code |
|---|---|---|
| 10 mg | $212.00 | D539-10 |
Product Description of Succinimidyl Alkanedisulfides
Succinimidyl ester-terminated alkyldisulfides are amine-reactive analogs of carboxyalkyldisulfide. They are utilized for the modification of a gold surface to introduce amine-reactive sites on the surface. It is possible to use this technique for protein chips and various sensors. There is no need to use coupling agents because these compounds are already activated. Wagner and others characterized dithiobis(succinimidyl undecanoate) SAMs on a gold substrate by scanning tunneling microscopy (STM), radiolabeling and in situ AFM imaging. The densely packed and highly reactive surfaces allowed them to easily immobilize amino acids and proteins.
Structural Formula
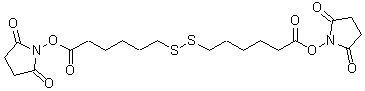
References
1) E. Delamarche, G. Sundarabaru, H. Biebuyck, B. Michel, Ch. Gerber, H. Sigrist, H. Wolf, H. Ringsdorf, N. Xanthopoulos and H. J. Mathieu, Immobilization of Antibodies on a Photoactive Self-Assembled Monolater on Gold, Langmuir, 1996, 12, 1997.
2) P. Wager, F. Zaugg, P. Kernen, M. Hegner and G. Semenza, "ω-functionalized self-assembled monolayers chemisorbed on ultraflat Au(111) surfaces for biological scanning probe microscopy in aqueous buffers", J. Vac. Sci. Technol. B, 1996, 14(2), 1466.
Handling and storage condition
| Appearance: | White powder |
|---|---|
| Purity (HPLC): | ≧ 90.0 % |
| NMR spectrum: | Authentic |
| 0-5°C, Nitrogen substitution, Protect from moisture |