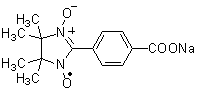
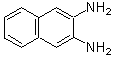
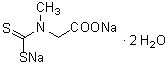BMPO
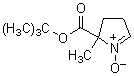
Spin Trap Reagent
- Long half-life
- High solubility in water
- Greater S/N ratio
-
Product codeB568 BMPO
-
CAS No.387334-31-8
-
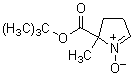
Chemical name5-tert-Butoxycarbonyl-5-methyl-1-pyrroline-N-oxide
-
MW199.25, C10H17NO3
| Unit size | Price | Item Code |
|---|---|---|
| 50 mg | $425.00 | B568-10 |
Description
Chemical Structure
Product Description
Spin trapping analysis is one of the most reliable techniques for detecting and identifying short-lived free radicals. The EPR (ESR) spin trap reagent detects both superoxide and hydroxyl radicals produced by systems in vitro and in vivo. BMPO was developed as a spin trapping reagent that adducts superoxide and shows a much longer half-life (t1/2=23 min) than other spin trap reagents. It gives us reproducible and steady results. Because BMPO is highly soluble in water, hydrophilic sample is applicable to analyze the free radicals.
Technical info
Measuring hydroxyl radical from Fenton reaction
1. Dissolve 1.5 mg of BMPO with 5 ml of ddH2O.
2. Add 15 μl of the BMPO solution, 75 μl of 1 mM H2O2 and 75 μl of 100 μM FeSO4 to 50 μl of ddH2O.
3. Transfer the solution to ESR sample tube and measure ESR spectra after certain time of period, e.g. 1 minute.
4. Calculate relative intensity from the peak height.
Measuring superoxide radical from xanthine oxidase(XO) reaction
1. Dissolve 1 mg of BMPO with 1 ml of 50 mM Phosphate buffer(pH7.4).
2. Prepare 50 mM Phosphate buffer(pH 7.4) containing 1 mM DTPA and 0.4 mM Xanthine.
3. Prepare 50 mM Phosphate buffer(pH 7.4) containing 0.1 U/ml xanthine oxidase.
4. Mix 15 μl of Solution A, 135 μl of solution B and 10 μl of Solution C.
5. Transfer the solution to ESR sample tube and measure ESR spectra after certain time of period, e.g. 8 minutes.
6. Calculate relative intensity from the peak height.
Superoxide Detection
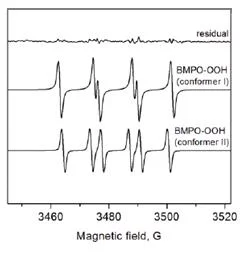
Protocol
1. Prepare a solution of 100 mM phosphate buffer (pH 7.4) containing 25 µM diethylenetriaminepentaacetic acid (DTPA) as transition metal chelator.
2. Make up a solution of 1 mM hypoxanthine in 100 mM phosphate buffer, pH 7.4.
3. Make up a solution of xanthine oxidase with concentration of 1 unit/ml
4. Make up a solution of BMPO with dissolved 10 mg of BMPO into 200 µl phosphate buffer (the final concentration should be 250 mM).
5. Prepare your reaction mixture to a total reaction volume of 200 µl.
6. Add 70 µl of buffer to an Eppendorf tube.
7. Add 20 µl of your 250 mM BMPO solution and 100 µl hypoxanthine of the stock 1 mM solution.
Hydroxyl radical detection
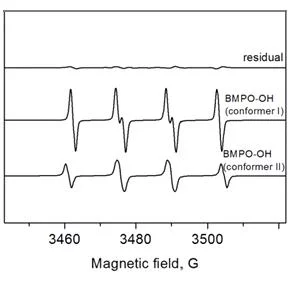
Protocol
1. Make up a solution of 1 mM FeSO4, 10 mM H2O2, and 250 mM BMPO in water.
2. Prepare your reaction mixture to a total reaction volume of 200 µl.
3. Add 140 µl of distilled water to an Eppendorf tube.
4. Add 20 µl of your 250 mM BMPO solution and 20 µl FeSO4 of the stock 1 mM solution.
5. Initiate the reaction with 20 µl 10 mM H2O2.
6. Mix the reactants and quickly transfer the solution to a flat cell.
7. Insert the flat cell into the cavity, tune the spectrometer, and acquire the spectrum.
The final concentrations of the components are: 25 mM BMPO, 0.1 mM FeSO4, and 1 mM H2O2.
Output spectra of the BMPO spin trapping of free hydroxyl radical are presented. In the case of BMPO, two stereoisomers of BMPO/OH were formed similar to BMPO/OOH results. The two BMPO adducts were fitted with aN = 13.47 G, aHβ = 15.31 G, and aHγ1 = 0.62 G for conformer I and aN = 13.56 G, aHβ = 12.3 G, and aHγ1 = 0.66 G for conformer II.
You should always perform control experiments in which one or more of the reagents are excluded. These experiments will reveal any paramagnetic impurities and will demonstrate that all the components were required to produce the EPR signal.
Time Course of the Superoxide Signal
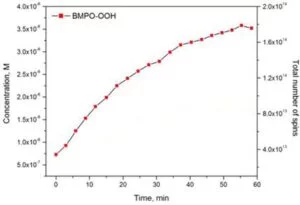
Data and Protocol were kindly provided by Bruker Corporation.
After feeding the results of SpinFit into SpinCount, the concentration changes over the time of the experiment are obtained.
Stability and Reaction Data for O2.- Detection
BMPO gives the rate constants for the reaction superoxide under different experimental conditions and longer stability of the corresponding spin trap adduct.
Link to Published Paper
References
Current Publications Using This Product
1) Tresp, H., Hammer, M. U., Winter, J., Weltmann, K. D., Reuter, S.; 2013; Quantitative detection of plasma-generated radicals in liquids by electron paramagnetic resonance spectroscopy; DOI:10.1088/0022-3727/46/43/435401
2) J Winter, H Tresp, M U Hammer, S Iseni, S Kupsch, A Schmidt-Bleker, K Wende, M Dünnbier, K Masur, K-D Weltmann, S Reuter; 2014; Tracking plasma generated H2O2 from gas into liquid phase and revealing its dominant impact on human skin cells; DOI:10.1088/0022-3727/47/28/285401
References
1) H. Zhao, J. Joseph, H. Zhang, H. Karoui and B. Kalyanaraman, Synthesis and biochemical applications of a solid cyclic nitrone spin trap: a relatively superior trap for detecting superoxide anions and glutathiyl radicals. Free Radic Biol Med. 2001;31:599-606.
2) G.M. Rosen, P. Tsai, J. Weaver, S. Porasuphatana, L. Roman, A. A. Starkov, G. Fiskum and S. Pou, The Role of Tetrahydrobiopterin in the Regulation of Neuronal Nitric-oxide Synthase-generated Superoxide. J Biol Chem. 2002;277:40275-40280.
Handling and storage condition
| Appearance: | White to slightly yellowish white crystal or crystalline powder |
|---|---|
| Purity: | ≥99.0% |
| -20°C, protect from moisture |