Arsemate
Metal Indicator
-
Product codeA012 Arsemate
-
CAS No.1470-61-7
-
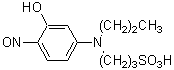
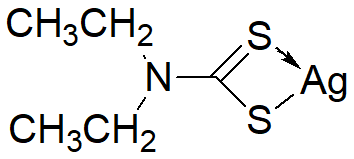
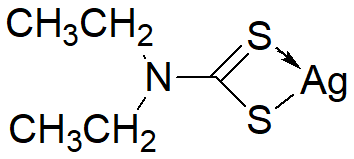
Chemical nameDiethyldithiOcarbamic acid, silver salt
-
MWC10AgNS2=256.14
| Unit size | Price | Item Code |
|---|---|---|
| 5 g | $70.00 | A012-10 |
| 25 g | $233.00 | A012-12 |
Chemical Structure
Product Description
Arsemate is soluble in pyridine and chloroform, but insoluble in water and other organic solvents. For arsenic detection, a 0.5% Arsemate/pyridine solution is used. The sample should be treated with sulfuric acid, 15% potassium iodide, and 40% SnCl2/HCl solution to convert arsenic to AsH3. Arsemate reacts with AsH3 in pyridine and forms red colloidal silver with a maximum wavelength at 520-540 nm. The detection range of arsenic using the Arsemate/pyridine solution is 4-12 ppm. Alternatively, 0.5% Arsemate/chloroform solution with 30 mM triethylamine can be used for the assay. For the colorimetric detection of arsenic, the AsH3 gas generator is required.
References
1. A. G. Howard and H. Arbab-Zavar, "Sequential Spectrophotometric Determination of Inorganic Arsenic(III) and Arcenic(V) Species", Analyst, 1980, 105, 33.
Handling and storage condition
| Appearance: | Slightly yellowish white to yellow powder or crystalline powder |
|---|---|
| Purity (Titration): | ≧ 98.0 % |
| Solubility in Pyridine: | To pass test (clear, yellow) |
| Solubility in Chloroform: | To pass test (clear, yellow) |
| Sensitivity (As): | To pass test |
| IR spectrum: | Authentic |
| Ambient temperature, Protect from light |