General Information
Singlet oxygen (1O2) is one of the Reactive Oxygen Species (ROS). 1O2 is known to be a cause of spots and wrinkles of the skin due to its very strong oxidizing potential. In the field of cancer research, 1O2 is of a particular importance because of its key role in photodynamic therapy (PDT), an emerging anticancer treatment using photoirradiation and photosensitizers. Therefore, the monitoring of 1O2 in living cells is highly important for understanding of anti-cancer mechanism of PDT. However, the existing fluorescence probe for the detection of 1O2 cannot be used in living cells because of its cell membrane impermeability.
Majima et al. synthesized a new far-red fluorescence probe composed of silicon-containing rhodamine and anthracene moieties, namely Si-DMA, as a fluorophore and a 1O2 reactive site, respectively. In the presence of 1O2, fluorescence of Si-DMA increases due to endoperoxide formation at the anthracene moiety1). Among seven different ROS, Si-DMA is able to selectively detect 1O2 (Fig. 3). In addition, Si-DMA is able to visualize the generation of 1O2 from protoporphyrin IX in mitochondria with 5-aminolevulinic acid (5-ALA), a precursor of heme (Fig. 4).
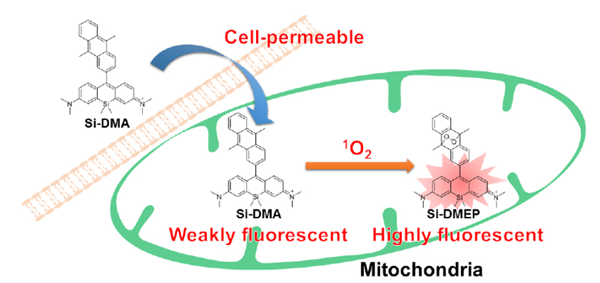
Fig.1 Cell staining mechanism by Si-DMA
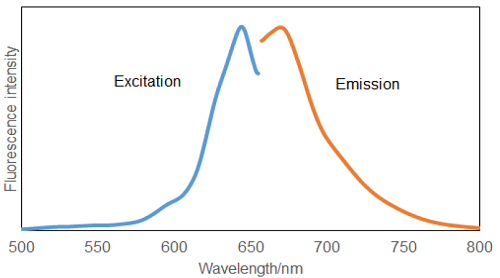
Fig.2 Excitation and Emission spectra of Si-DMA after reaction with 1O2
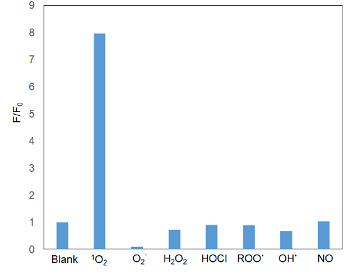
Fig.3 Selectivity of Si-DMA toward various ROS
Kit Contents
| Si-DMA | 2 µg x 1 |
Storage Condition
Store at -20 ℃ and protect from light.
- Si-DMA is sensitive to light. Store unused Si-DMA in the bag at -20 ℃.
Required Equipment and Materials
- Dimethyl sulfoxide (DMSO)
- Hanks’ HEPES buffer or HBSS
- Micropipettes
Preparation of Solutions
Preparation of 100 μmol/l Si-DMA DMSO stock solution
Add 36 μl of DMSO to a tube of Si-DMA (2 μg) and dissolve it with pipetting.
- Store the Si-DMA DMSO stock solution at -20 ℃ .
Si-DMA DMSO stock solution is stable at -20 ℃ for up to a month.
Preparation of Si-DMA working solution
Dilute the Si-DMA DMSO stock solution with Hanks’ HEPES buffer to prepare 25-100 nmol/l Si-DMA working solution.
- Protect the solution from light and use it within the same day, since Si-DMA is not stable in Hanks’ HEPES buffer.
Notes
The recommended concentration of Si-DMA working solution for use is 25-100 nmol/l. Once the final concentration of Si-DMA becomes more than 1 μmol/l, Si-DMA may be accumulated to organelle. In addition, the final concentration of Si-DMA becomes more than 5 μmol/l, cytotoxicity may be seen.
General Protocol
Si-DMA staining
- Prepare cells for the assay.
- Discard the culture medium and wash the cells with Hanks’ HEPES buffer twice.
- Add an appropriate volume of Si-DMA working solution.
- Incubate for 45 minutes at 37 ℃.
- Discard the supernatant and wash the cells with Hanks’ HEPES buffer twice.
- Add Hanks’ HEPES buffer and observe the cells under a fluorescence microscope.
Usage Examples
Fluorescence microscopic detection of 1O2 in HeLa cells treated with 5-aminolevulinic acid (5-ALA)
- HeLa cells at 2.4×105 cells/ml (200 μl) were seeded on a μ-slide 8 well (Ibidi) in DMEM (10% fetal bovine serum, 1% penicilin-streptmycin) and cultured at in a 5% CO2 incubator overnight at 37 ℃.
- The cells were washed with 200 μl of Hanks’ HEPES buffer twice.
- 5-ALA in Hanks’ HEPES buffer (150 μg/ml, 200 μl) was added to the μ-slide, and the cells were cultured in a 5% CO2 incubator for 4 hours at 37 ℃.
- The cells were washed with 200 μl of Hanks’ HEPES buffer twice.
- Si-DMA working solution (40 nmol/l, 200 μl) was added, and the cells were cultured in a 5% CO2 incubator for 45 minutes at 37 ℃.
- The cells were washed with 200 μl of Hanks’ HEPES buffer twice.
- Hanks’ HEPES buffer (200 μl) were added, and the cells were observed under a fluorescence microscope.
Filter (wavelength/band pass)
Fluorescence imaging: 600/50 nm (Ex), 685/50 nm (Em)
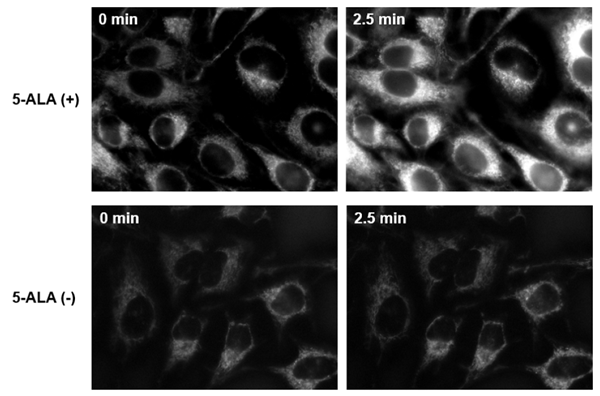
Fig.4 Fluorescence imaging of mitochondrial 1O2 with Si-DMA in HeLa cells treated with 5-ALA.
Fluorescent of Si-DMA in 5-ALA-treated HeLa cells increased after 2.5 minutes irradiation. It was found that Si-DMA was able to visualize in real time the 1O2 generated from protoporphyrin IX in mitochondria.
References
- S. Kim, T. Tachikawa, M. Fujitsuka, T. Majima, “Far-Red Fluorescence Probe for Monitoring Singlet Oxygen
during Photodynamic Therapy”, J. Am. Chem. Soc., 2014, 136 (33), 11707-11715.
Frequently Asked Questions / Reference
MT05: Si-DMA for Mitochondrial Singlet Oxygen Imaging
Revised Aug., 30, 2023


 Hidden sections will not be printed.
Hidden sections will not be printed.

