General Information
The lysosome is an organelle in which an acid vacuole is formed by a biomembrane. Lysosomes contain various degrading enzymes and contribute to maintaining intracellular homeostasis by acting as a waste disposal system. Recent findings reveal that lysosomal dysfunction is related to some neurodegenerative disorders. Consequently, investigation of lysosomal function is attracting considerable interest in the scientific community.
Many types of small fluorescent probes are used for monitoring lysosomes in living cells. Dojindo’s LysoPrime Green overcomes known problems with fluorescent lysosome probes, such as lack of specificity for lysosomes and staining dependent on the lysosomal pH. In addition, the high-retentivity of LysoPrime Green enables long-term imaging experiments.
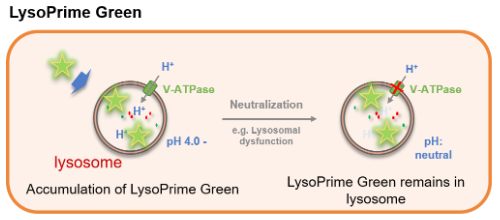
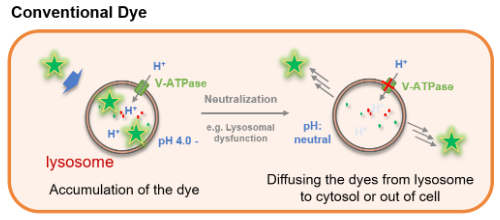
Figure 1. The difference between LysoPrime Green and conventional lysosome staining dye
Fluorescent Property
Excitation and emission spectra of LysoPrime Green
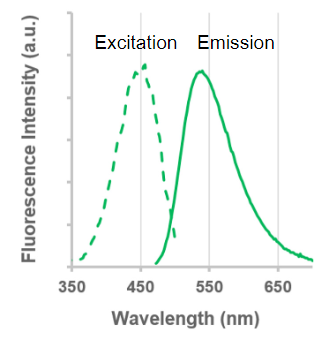 |
λex: 456 nm recommended filter settings |
Contents
| LysoPrime Green | 10 μl x 1 (corresponds to 10 dishes (35 mm) ) 10 μl x 3 (corresponds to 30 dishes (35 mm) ) |
Storage Condition
Store at -80 °C
- The influence of storage temperature on the staining ability of LysoPrime Green is described in the FAQ on our website (product code: L261).
Required Equipment and Materials
- Growth medium
- Hanks’ Balanced Salt Solution (HBSS) or serum free medium
- Micropipettes
- Microtubes
Preparation of Solution
Preparation of LysoPrime Green working solution
- Dilute the LysoPrime Green solution 2000 times with HBSS or serum free medium to prepare LysoPrime Green working solution.
- The final concentration of LysoPrime Green should be optimized depending on the cell line (dilution range: 1000 – 4000 times). Use HBSS or serum-free medium for the dilution because serum in medium interferes with LysoPrime Green. LysoPrime Green working solution should be used on the day it is prepared.
General Protocol
- Seed cells in a dish and culture them overnight at 37 °C in an incubator equilibrated with 95% air and 5% CO2.
- Discard the culture medium and wash the cells once with HBSS or serum free medium.
- Add LysoPrime Green working solution to the dish containing the cells and incubate them for 30 minutes at 37 °C in an incubator equilibrated with 95% air and 5% CO2.
- Discard the supernatant and wash the cells twice with HBSS or serum free medium.
- Add growth medium to the dish, then observe the cells under a fluorescence microscope.
Usage Example
Observation of the lysosomal pH change in HeLa cells
- HeLa cells in MEM (containing 10 % fetal bovine serum) were seeded (1.0 × 104 cells/well) on a μ-slide 8 well plate (ibidi) and cultured overnight at 37 °C in an incubator equilibrated with 95% air and 5% CO2.
- After washing once with HBSS, 200 μl of working solution [LysoPrime Green: 2000 times dilution, LysoTracker Red (LTR): 100 nmol/l] was added to the plate, and the cells were incubated at 37 °C for 30 min.
- The cells were washed twice with HBSS.
- HBSS (200 μl) containing 100 nmol/l Bafilomycin A1, an inhibitor of lysosomal acidification, was added to the plate, and the cells were incubated at 37 °C for 30 min.
- The cells were washed twice with PBS.
- MEM was added to the plate, and the cells were observed under a confocal fluorescence microscope.
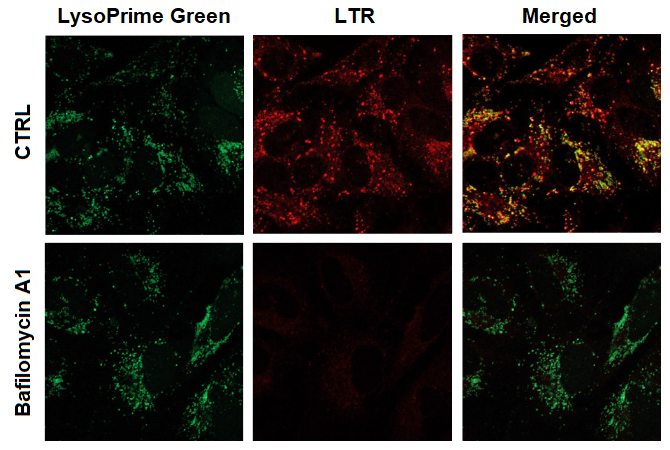
Figure 2. The effect of Bafilomycin A1 on lysosomal pH
| CTRL: Normal condition, Bafilomycin A1: Inhibition of lysosomal acidification LysoPrime Green filter sets: 488 nm (Ex), 500 – 570 nm (Em) LysoTracker Red (LTR) filter sets: 548 nm (Ex), 550 – 650 nm (Em) |
Frequently Asked Questions / Reference
L261: LysoPrime Green - High Specificity and pH Resistance
Revised Mar., 21, 2023


 Hidden sections will not be printed.
Hidden sections will not be printed.

