|
Lysosome dependent degradation in neurons and surrounding glial cells is essential for maintaining neural homeostasis under stress. Clarifying how these pathways process protein aggregates and lipid burden is important for understanding mechanisms of neuronal protection and intercellular responses in the nervous system. Recent studies showed that exposure of neurons to aggregated α-synuclein impairs lysosomal and autophagic function, thereby promoting tunneling nanotube mediated transfer of aggregates to microglia. Another study showed that the endolysosomal protein TTYH1 supports astrocytic autophagic flux and lipid droplet degradation under inflammatory stress, contributing to metabolic adaptation to neuron derived lipid signals. Together, these findings identify glial lysosome related pathways as one component of neuron protective responses. |
||||||||||||||||||||||
|
Summary: Aggregated α-synuclein is a key feature of neurodegenerative diseases and can transfer from neurons to microglia through tunneling nanotubes. This study demonstrates that impaired autophagy in neurons reduces their ability to degrade α-synuclein aggregates, thereby promoting their TNT-mediated transfer to microglia. It further reveals that microglia possess higher lysosomal turnover and efficiently clear the transferred aggregates. Altogether, the findings identify dysfunctional neuronal autophagy as a central driver of the directional spread of α-synuclein aggregates toward microglia. Highlighted technique: To determine whether differences in degradative capacity between neurons and microglia drive the directional transfer of α-synuclein aggregates, the authors exposed each cell type to α-synuclein aggregates and assessed their intracellular handling using AlexaFluor647-conjugated Dextran for lysosome tracking and DQ-BSA for lysosomal degradative activity measurements. In addition, they evaluated autophagic flux using p62 puncta, which can reflect either increased autophagosome formation or impaired cargo degradation, in the presence or absence of bafilomycin A1. |
||||||||||||||||||||||
|
Summary: Neuronal lipid handling depends in part on surrounding astrocytes, and this study shows that the endolysosomal protein TTYH1 is a key factor supporting autophagic flux and lipid droplet degradation in astrocytes. Under inflammatory stress, neuron-derived ceramide 1-phosphate signaling places a burden on these astrocytic endolysosome-dependent degradative pathways, which is alleviated by TTYH1-dependent endolysosomal clearance. Highlighted technique: The authors used primary astrocytes expressing the tandem reporter GFP-LC3-RFP-LC3ΔG to monitor autophagic flux under starvation conditions in the presence or absence of TTYH1. In this reporter, GFP-LC3 is degraded by autophagy whereas RFP-LC3ΔG remains in the cytosol as an internal control, allowing the GFP/RFP signal ratio to be used as a readout of autophagic flux. |
||||||||||||||||||||||
|
Solutions for Lysosome Experiments |
||||||||||||||||||||||
All Related Techniques (click to open/close)
|
||||||||||||||||||||||
Application Note (click to open/close)
|
||||||||||||||||||||||
|
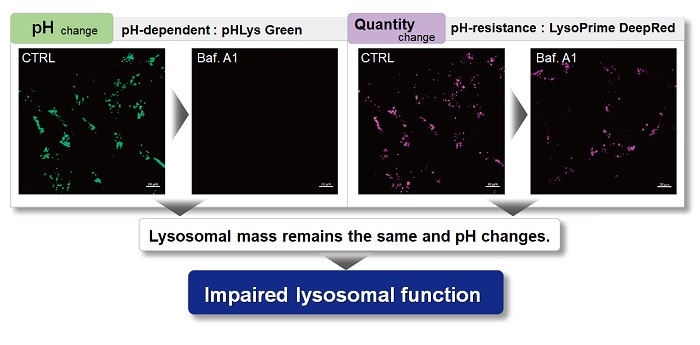
With existing reagents, it was difficult to determine whether lysosomal mass or their function (pH) fluctuated because the discussion was based on changes in the fluorescence brightness of a single dye. This kit contains pHLys Green, which is highly specific to lysosomes and shows pH-dependent changes in fluorescence, and pH-resistant LysoPrime Deep Red. Using these two dyes, lysosomal pH and volume of the same sample can be measured for a detailed analysis of lysosomal function.
|
|||
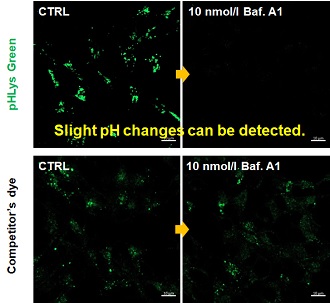
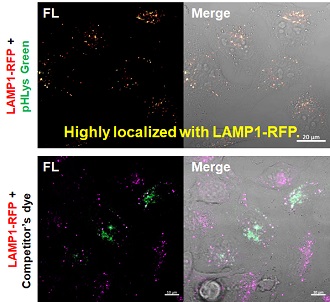
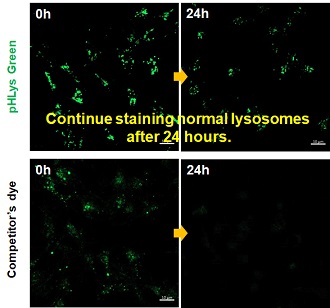
| Existing lysosomal pH detection reagents have issues with dye localization, pH sensitivity, and retention. pHLys Green is a dye that solves these issues. The improved dye retention and localization enable detection of normal lysosomes, and the improved pH sensitivity enables detection of slight pH changes. | |||
| 1. High sensitive pH detection Comparison of pH response of cells treated with low concentrations of lysosomal acidification inhibitor Bafilomycin A1 |
2. High specificity for lysosomes Comparison of specificity for lysosomes using lysosomal marker protein LAMP1-GFP expressing cells |
3. High retention in lysosomes Comparison of intracellular retention |
|
 |
 |
 |
|
|
Product in Use: Related Product: |
|||