|
Neuronal homeostasis depends on lysosomal function to maintain metabolic and redox balance. Lysosomal dysfunction is frequently associated with neurological disease. Recent work shows that loss of the neuronal lysosomal sugar transporter SLC45A1 causes lysosomal sugar accumulation, reduced V-ATPase V1 abundance and acidification, iron and ROS imbalance, and impaired mitochondrial respiration. Another study identifies lysosomal SLC7A11 (xCT) as a cystine/glutamate exchanger that regulates lysosomal pH. Its inhibition triggers hyperacidification, impaired degradation, ferroptosis, and alpha-synuclein aggregation. These results link defined lysosomal transport processes to neuronal homeostasis and disease-associated phenotypes. |
||||||||||||||||||||||
|
Summary: SLC45A1 mutations cause a monogenic neurological disorder, yet its neuronal function was unclear. This study shows SLC45A1 is a neuronal lysosomal sugar transporter, and its loss causes lysosomal sugar accumulation, reduced V-ATPase V1 and acidification, iron and ROS imbalance, and impaired mitochondrial respiration, explaining how lysosomal failure and metabolic stress may contribute to neurological symptoms. Highlighted technique: To test whether SLC45A1 loss disrupts lysosomal homeostasis and downstream metabolic stress, the authors compared control, SLC45A1-deficient, and rescued cells and quantified lysosomal pH using a ratiometric pH assay. They then measured ROS with a fluorescent probe, assessed cellular iron status by ferritin immunoblotting, and linked these changes to mitochondrial dysfunction by Seahorse OCR analysis. |
||||||||||||||||||||||
|
2. SLC7A11 is an unconventional H+ transporter in lysosomes (Cell, 2025) Summary: This study identifies lysosomal xCT (SLC7A11) as a previously unrecognized mediator of slow proton leak via cystine/glutamate exchange, revealing a novel mechanism for regulating lysosomal pH. Inhibition of SLC7A11 leads to lysosomal hyper acidification, impaired degradation, ferroptosis, and α-synuclein aggregation. Highlighted technique: The authors screened a lysosomal membrane protein KO library by measuring lysosomal acidity with a pH-sensitive dye. Unlike most cells, SLC7A11-KO cells maintained acidity after bafilomycin A1 treatment, identifying SLC7A11 as a key regulator of lysosomal H⁺ efflux. |
||||||||||||||||||||||
|
Solutions for Lysosome Experiments |
||||||||||||||||||||||
All Related Techniques (click to open/close)
|
||||||||||||||||||||||
Application Note (click to open/close)
|
||||||||||||||||||||||
|
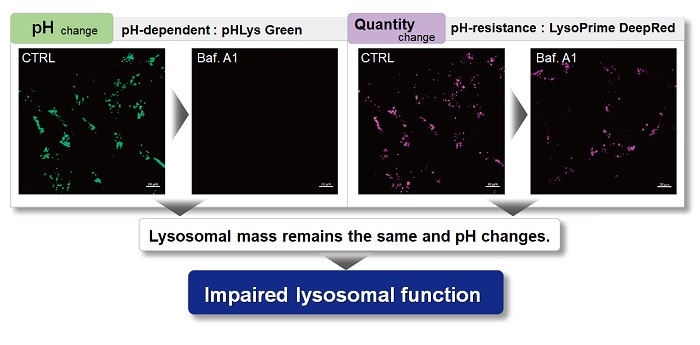
With existing reagents, it was difficult to determine whether lysosomal mass or their function (pH) fluctuated because the discussion was based on changes in the fluorescence brightness of a single dye. This kit contains pHLys Green, which is highly specific to lysosomes and shows pH-dependent changes in fluorescence, and pH-resistant LysoPrime Deep Red. Using these two dyes, lysosomal pH and volume of the same sample can be measured for a detailed analysis of lysosomal function.
|
|||
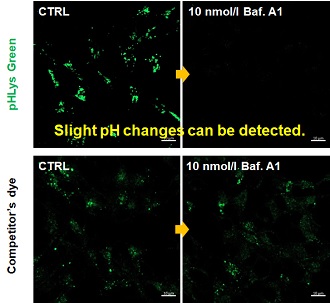
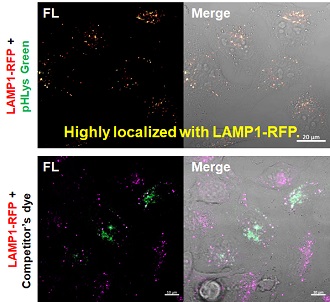
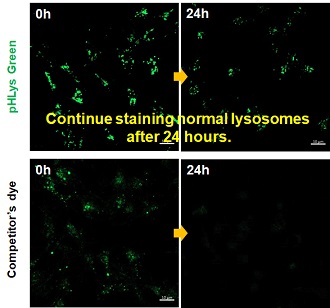
| Existing lysosomal pH detection reagents have issues with dye localization, pH sensitivity, and retention. pHLys Green is a dye that solves these issues. The improved dye retention and localization enable detection of normal lysosomes, and the improved pH sensitivity enables detection of slight pH changes. | |||
| 1. High sensitive pH detection Comparison of pH response of cells treated with low concentrations of lysosomal acidification inhibitor Bafilomycin A1 |
2. High specificity for lysosomes Comparison of specificity for lysosomes using lysosomal marker protein LAMP1-GFP expressing cells |
3. High retention in lysosomes Comparison of intracellular retention |
|
 |
 |
 |
|
|
Product in Use: Related Product: |
|||