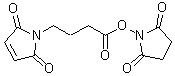
GMBS
Cross-Linker
-
Product codeG005 GMBS
-
CAS No.80307-12-6
-
Chemical name340-08951
-
MWC12H12N2O6=280.23
| Unit size | Price | FUJIFILM Wako Products code |
|---|---|---|
| 50 mg | ¥12,300 | 347-05041 |
| 100 mg | ¥20,700 | 343-05043 |
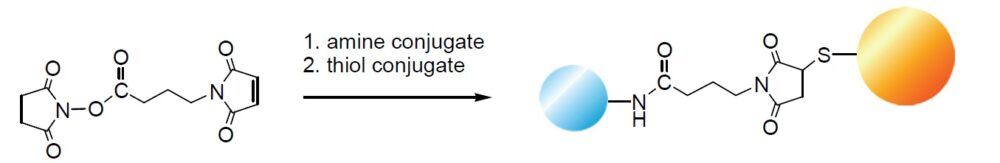
Cross-linking Reaction
Product Description
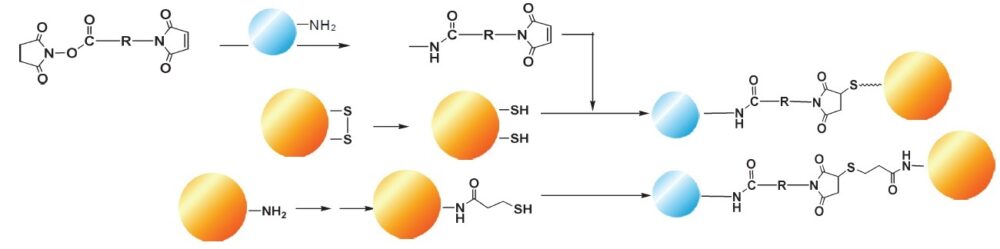
Hetero-bifunctional cross-linking reagents have activated esters and maleimide reactive groups. These functional groups react with amines and sulfhydryl groups of proteins, respectively. Enzyme-labeled haptens are prepared using hetero-bifunctional cross-linking reagents such as EMCS or GMBS. The cross-linking reaction requires neutral pH and mild temperature because it is necessary to maintain enzyme activity and antibody titers in the cross-linking reaction. Hetero-bifunctional cross-linking reagents with 3, 5, 7, or 10 linear carbon chains are available. These linear aliphatic chains act as spacers between the two reactive sites and their water-soluble reagents. They are more stable than the aromatic cross-linking reagents such as succinimidyl-4-N-maleimidobenzoate in a wider pH range.
Conjugation of Macromolecules with Hetero-Bifunctional Cross-Linking Reagent
Manual
Technical info
References
1. S. Yoshitake, Y. Yamada, E. Ishikawa and Rene Masseyeff, Conjugation of Glucose Oxidase from Aspergillus Niger and Rabbit Antibodies Using N-Hydroxysuccinimide Ester of N-(4-Carboxycyclohexylmethyl)-Maleimide, Eur. J. Biochem., 1979, 101, 395.
2. H. Tanimori, T. Kitagawa, T. Tsunoda and R. Tsuchiya, Enzyme Immunoassay of Neocarzinostain Using β-Galactosidase as Label, J. Pharm. Dyn., 1981, 4, 812.
3. K. Fujiwara, M. Yasuno and T. Kitagawa, Novel Preparation Method of Immunogen for Hydrophobic Hapten, Enzyme Immunoassay for Daunomycin and Adriamycin, J. Immunol. Methods, 1981, 45, 195.
4. T. Miura, H. Kouno and T. Kitagawa, Detection of Residual Penicillin in Milk by Sensitive Enzyme Immunoassay, J. Pharm. Dyn., 1981, 4, 706.
5. S. Yoshitake, M. Imagawa, E. Ishikawa, Y. Niitsu, I. Urushizaki, M. Nishiura, R. Kanazawa and H. Ogawa, Mild and Efficient Conjugation of Rabbit Fab Eand Horseradish Peroxidase Using a Maleimide Compound and Its Use for Enzyme Immunoassay, J. Biochem. ,1982, 92, 1413.
6. T. Kitagawa, H. Tanimori, K. Yoshida, H. Asada, T. Miura and K. Fujiwara, Studies on Viomycin. XV. Comparative Study on the Specificities of Two Anti-viomycin Antisera by Enzyme Immunoassay, Chem. Pharm. Bull., 1982, 30, 2487.
7. T. Kitagawa, T. Kawasaki and H. Munechika, Enzyme Immunoassay of Blasticidin S with High Sensitivity:A New and Convenient Method for Preparation of Immunogenic(Hapten-Protein) Conjugates, J. Biochem.,1982, 92, 585.
Handling and storage condition
| Appearance: | White powder |
|---|---|
| Purity (HPLC): | ≧95.0% (HPLC) |
| Solubility in Chloroform: | To pass test (clear, colorless) (50 mg / 5ml) |
| Solubility in Dimethylformamide: | To pass test (clear, colorless) (50 mg / 5ml) |
| m.p.: | 124 - 132℃ |
| IR spectrum: | Authentic |
| NMR spectrum: | Authentic |
| 0-5°C |









