| Mitochondrial Ca2+ control in neurons links activity to metabolism and is important for neuronal function and memory. Defining the proteins that regulate mitochondrial Ca2+ flux is therefore important for understanding cognitive decline and neurodegeneration. Recent studies showed that loss of the mitochondrial H+/Ca2+ exchanger Letm1 reduces Ca2+ efflux, thereby sustaining mitochondrial Ca2+ signals, extending metabolic activation, and enhancing long term memory in flies and mice. Another study showed that TMEM65 is necessary and sufficient for mitochondrial Na+/Ca2+ exchange, thereby identifying the molecular basis of mito-NCX, a major neuronal mitochondrial Ca2+ efflux pathway. Together, these findings clarify major regulators of neuronal mitochondrial Ca2+ efflux relevant to dementia related biology. | ||||||||||||||||||||||||
|
Mitochondrial Ca2+ efflux controls neuronal metabolism and long-term memory across species (Nature Metabolism, 2026) |
||||||||||||||||||||||||
|
TMEM65 functions as the mitochondrial Na+/Ca2+ exchanger (Nature Cell Biology, 2025) |
||||||||||||||||||||||||
Mitochondrial and Metabolic Activity Indicators (click to open/close)
|
||||||||||||||||||||||||
Application Note I (click to open/close)
|
||||||||||||||||||||||||
|
|
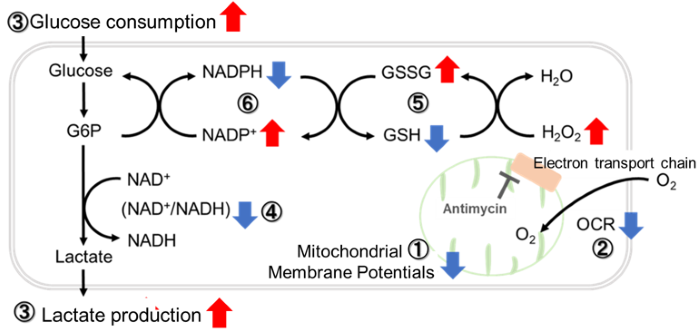
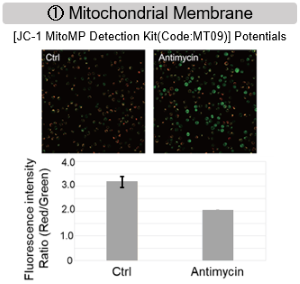
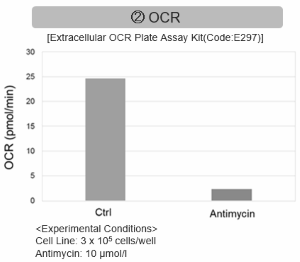
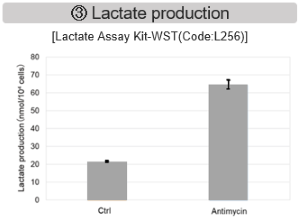
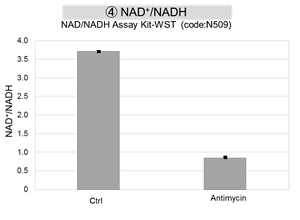
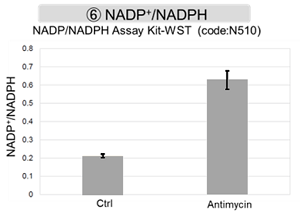
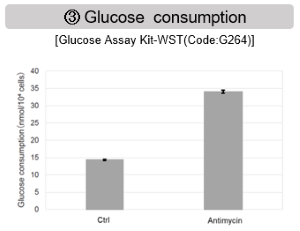
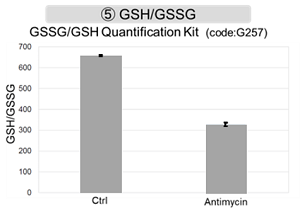
Antimycin stimulation of Jurkat cells was used to evaluate the changes in cellular state upon inhibition of the mitochondrial electron transport chain using a variety of indicators. The results showed that inhibition of the electron transport chain resulted in (1) a decrease in mitochondrial membrane potential and (2) a decrease in OCR. In addition, (3) the NAD+/NADH ratio of the entire glycolytic pathway decreased due to increased metabolism of pyruvate to lactate to maintain the glycolytic pathway, (4) GSH depletion due to increased reactive oxygen species (ROS), and (6) increase in the NADP+/NADPH ratio due to decreased NADH required for glutathione biosynthesis were observed.
|
Application Note II (click to open/close)
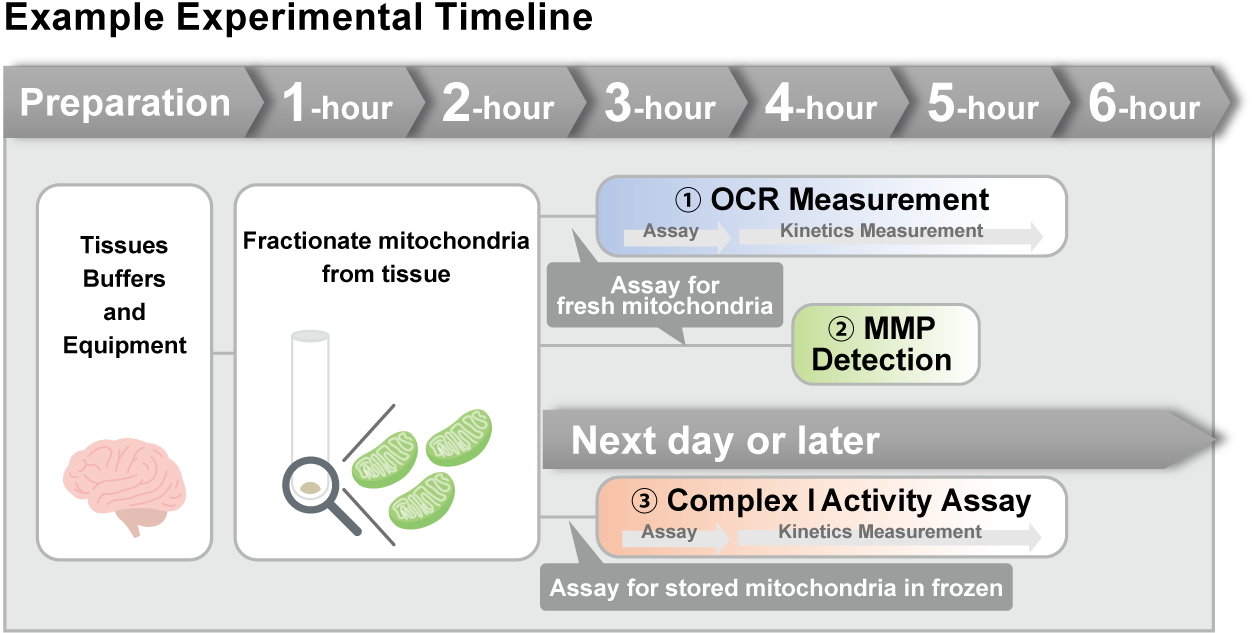
> Activity Evaluation of Mitochondria Fractionated from Mouse Brain
|
|
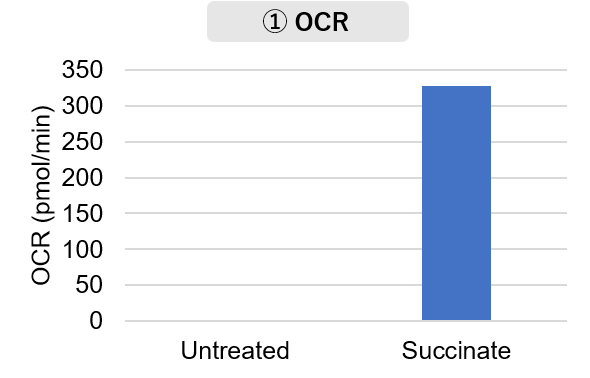
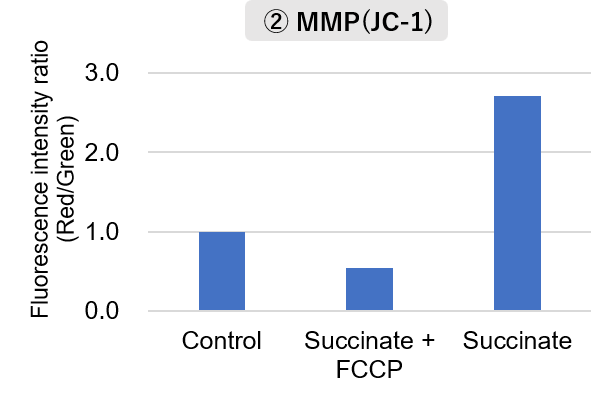
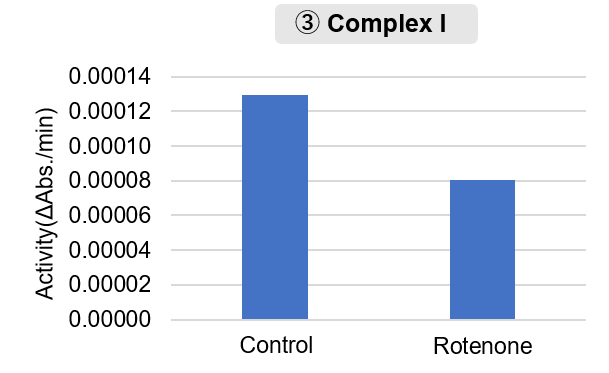
Mitochondria were isolated from mouse brain tissue, and oxygen consumption rate (OCR), mitochondrial membrane potential (MMP), and Complex I activity were measured. The results showed that the addition of succinate, a substrate that activates Complex II of the electron transport chain, increased both OCR and MMP. In contrast, FCCP treatment reduced MMP, indicating that intact mitochondria were successfully fractionated. |
   |
|
| <Product used> Mitochondrial Fractionation: IntactMito Fractionation Kit for Tissue (Code: MT17) OCR measurement: Extracellular OCR Plate Assay Kit (Code: E297) MMP detection: JC-1 MitoMP Detection Kit (Code: MT09) Complex I activity assay: MitoComplex- I Activity Assay Kit (Code: MT18) |
<Experimental Conditions> |