-SulfoBiotics- Sodium disulfide (Na2S2)
Biosulfur Analysis
-
Product codeSB02 -SulfoBiotics- Sodium disulfide (Na2S2)
-
CAS No.22868-13-9
-
Chemical nameSodium disulfide, anhydrous
-
MWNa2S2=110.11
| Unit size | Price | Item Code |
|---|---|---|
| 100 mg x 5 | $168.00 | SB02-10 |
Product Description:
It becomes obvious that there are a lot of molecules containing sulfane sulfurs such as persulfides and polysulfides in living organism. These molecular species are involved in production, storage, and release of hydrogen sulfide, which is recognized as an important physiological mediator. Furthermore, recent studies reveal that persulfides and polysulfides may control intracellular signal transduction through s-sulfhydration of proteins, and function in vivo as anti-oxidants which have much higher reducing activity than glutathione in reduced form or cysteine.
Sodium polysulfides (Na2Sn) are sulfane sulfur donors which have simple structures, and exist as hydrogen polysulfides in an aqueous solution depending on pH. These reagents are necessary to for research and analysis of sulfane sulfurs in vivo.
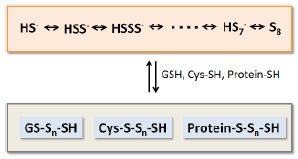
Fig. 1 Chemical species containing sulfane sulfurs
Characteristics of polysulfides
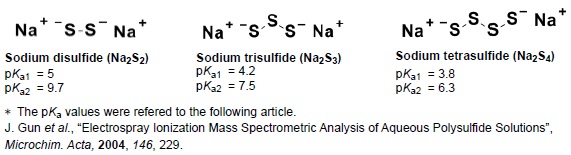
Fig. 2 Structures of Sodium polysulfides (Na 2Sn) and the pKa values.
| Developer | 開発元:Dojindo Molecular Technologies, Inc. |
|---|
Manual
References
1. Y. Kimura, Y. Mikami, K. Osumi, M. Tsugane, J. Oka, and H. Kimura, “Polysulfides are possible H 2S-derived signaling molecules in rat brain”, FASEB J., 2013, 27, 2451.
2. S. Koike, Y. Ogasawara, N. Shibuya, H. Kimura, and K. Ishii, “Polysulfide exerts a protective effect against cytotoxicity caused by t-buthylhydroperoxide through Nrf2 signaling in neuroblastoma cells”, FEBS Lett., 2013, 587, 3548.
Handling and storage condition
| Appearance: | Yellow to yellowish brown powder or solid |
|---|---|
| Purity: | ≧ 90.0 % |
| 0-5°C, Protect from light, Protect from moisture |










