Biotin Labeling Kit - SH
Antibody / Protein Labeling
- Only 3 hours to recover conjugates
- All processes in a single filtration tube
- High recovery of conjugates
- Applicable for 50-200 μg IgG
-
Product codeLK10 Biotin Labeling Kit - SH
| Unit size | Price | Item Code |
|---|---|---|
| 3 samples | $179.00 | LK10-10 |
| 3 samples | ・SH-Reactive Biotin ・Reducing Agent ・WS Buffer ・Reaction Buffer ・Filtration Tube |
3 tubes 3 tubes 4 ml x 1 1 ml x 1 3 tubes |
|---|
Description
Biotin Labeling Kit-SH is mainly used for the preparation of biotin-labeled IgG for enzyme immunoassay (EIA). SH reactive biotin, a component of this kit, reacts with sulfhydryl groups of proteins or other molecules (Fig.1). The kit contains all the reagents necessary for the labeling. The reducing agent included in this kit creates sulfhydryl groups in the IgG molecule. Because the reducing agent is a water-soluble phosphine compound, it can be removed by a filtration tube with a simple process. An average of 5 to 8 biotin molecules conjugate to each IgG molecule. The number of biotin molecules per protein can be determined by HABA assay. Excess biotin molecules can be removed by a filtration tube.
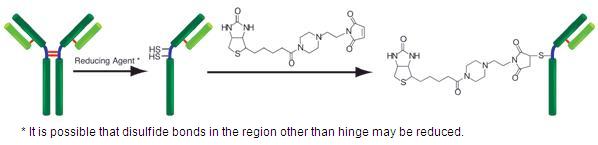
Fig. 1 IgG labeling reaction with SH-reactive biotin
Precaution
♦ The molecular weight of the reduced protein to be labeled with this kit should be greater than 50,000.
♦ IgG or biotin-conjugated IgG is always on the membrane of the filtration tube during the labeling process.
♦ If the IgG solution contains other proteins with molecular weights larger than 10,000, such as BSA or gelatin, purify the IgG solution before labeling biotin with this kit.
♦ If the IgG solution contains small insoluble materials, centrifuge the solution and use the supernatant for the labeling.
| Developer | Dojindo Molecular Technologies, Inc. |
|---|
Manual
References
1) E. Terasaka, K. Yamada, P.H. Wang, K. Hosokawa, R. Yamagiwa, K. Matsumoto, S. Ishii, T. Mori, K. Yagi, H. Sawai, H. Arai, H. Sugimoto, Y. Sugita, Y. Shiro and T. Tosha, "Dynamics of nitric oxide controlled by protein complex in bacterial system", Proc. Natl. Acad. Sci. U.S.A.., 2017, 114, (37), 9888.
2) J. Hiruma, K. Harada, A. Motoyama, Y. Okubo, T. Maeda, M. Yamamoto, M. Miyai, T. Hibino and R. Tsuboi, "Key component of inflammasome, NLRC4, was identified in the lesional epidermis of psoriatic patients", J. Dermatol.., 2018, 45, (8), 971.
3) K. Gonda, M. Watanabe, H. Tada, M. Miyashita, Y. Takahashi-Aoyama, T. Kamei, T. Ishida, S. Usami, H. Hirakawa, Y. Kakugawa, Y. Hamanaka, R. Yoshida, A. Furuta, H. Okada, H. Goda, H. Negishi, K. Takanashi, M. Takahashi, Y. Ozaki, Y. Yoshihara, Y. Nakano and N. Ohuchi, "Quantitative diagnostic imaging of cancer tissues by using phosphor-integrated dots with ultra-high brightness", Sci. Rep.., 2017, 7, (1), 7509.
4) K. Saito, M. Sakaguchi, H. Iioka, M. Matsui, H. Nakanishi, N.H. Huh and E. Kondo, "Coxsackie and adenovirus receptor is a critical regulator for the survival and growth of oral squamous carcinoma cells", Oncogene., 2014, 33, (10), 127401286.
5) K. Yamamoto, H. Murata, E.W. Putranto, K. Kataoka, A. Motoyama, T. Hibino, Y. Inoue, M. Sakaguchi and N.H. Huh, "DOCK7 is a critical regulator of the RAGE-Cdc42 signaling axis that induces formation of dendritic pseudopodia in human cancer cells", Oncol. Rep.., 2013, 29, (3), 1073.
6) M. Sakaguchi, H. Murata, K. Yamamoto, T. Ono, Y. Sakaguchi, A. Motoyama, T. Hibino, K. Kataoka and N.H. Huh, "TIRAP, an Adaptor Protein for TLR2/4, Transduces a Signal from RAGE Phosphorylated upon Ligand Binding", PLoS ONE., 2011, 6, (8), e23132.
7) M. Sakaguchi, H. Murata, Y. Aoyama, T. Hibino, E.W. Putranto, I.M. Ruma, Y. Inoue, Y. Sakaguchi, K. Yamamoto, R. Kinoshita, J. Futami, K. Kataoka, K. Iwatsuki and N.H. Huh, "DNAX-activating Protein 10 (DAP10) Membrane Adaptor Associates with Receptor for Advanced Glycation End Products (RAGE) and Modulates the RAGE-triggered Signaling Pathway in Human Keratinocytes", J. Biol. Chem.., 2014, 289, (34), 23389.
8) N. Kobayashi, K. Odaka, T. Uehara, K. Imanaka-Yoshida, Y. Kato, H. Oyama, H. Tadokoro, H. Akizawa, S. Tanada, M. Hiroe, T. Fukumura, I. Komuro, Y. Arano, T. Yoshida and T. Irie, "Toward in Vivo Imaging of Heart Disease Using a Radiolabeled Single-Chain Fv Fragment Targeting Tenascin-C", Anal. Chem.., 2011, 83, (23), 9123.
9) T. Ikeda, R. Shinohata, M. Murakami, K. Hina, S. Kamikawa, S. Hirohata, S. Kusachi, A. Tamura and S. Usui, "A rapid and precise method for measuring plasma apoE-rich HDL using polyethylene glycol and cation-exchange chromatography: a pilot study on the clinical significance of apoE-rich HDL measurements", Clin. Chim. Acta.,2017, 465, 112.
10) T. Into, M. Inomata, M. Nakashima, K. Shibata, H. Hacker and K. Matsushita, "Regulation of MyD88-Dependent Signaling Events by S Nitrosylation Retards Toll-Like Receptor Signal Transduction and Initiation of Acute-Phase Immune Responses", Mol. Cell. Biol.., 2008, 28, (4), 1338.
11) W. Nakai, T. Yoshida, D. Diez, Y. Miyatake, T. Nishibu, N. Imawaka, K. Naruse, Y. Sadamura and R. Hanayama, "A novel affinity-based method for the isolation of highly purified extracellular vesicles", Sci. Rep.., 2016, 6, 33935.
12) Y. Terasaki, T. Akuta, M. Terasaki, T. Sawa, T. Mori, T. Okamoto, M. Ozaki, M. Takeya and T. Akaike, "Guanine Nitration in Idiopathic Pulmonary Fibrosis and Its Implication for Carcinogenesis", Am. J. Respir. Crit. Care Med.., 2006, 174, (6), 665.
13) I. W. Sumardika, C. Youyi, E. Kondo, Y. Inoue, I. M. W. Ruma, H. Murata, R. Kinoshita, K. Yamamoto, S. Tomida, K. Shien, H. Sato, A. Yamauchi, J. Futami, E. W. Putranto, T. Hibino, S. Toyooka , M. Nishibori and M. Sakaguchi, "β-1,3-Galactosyl-O-Glycosyl-Glycoprotein β-1,6-N-Acetylglucosaminyltransferase 3 Increases MCAM Stability, Which Enhances S100A8/A9-Mediated Cancer Motility.", Oncol. Res., 2018, 26, (3), 431.
14) J. Iwano, D. Shinmi, K. Masuda, T. Murakami and J. Enokizono, "Impact of Different Selectivity between Soluble and Membrane-bound Forms of Carcinoembryonic Antigen (CEA) on the Target-mediated Disposition of Anti-CEA Monoclonal Antibodies.", Drug Metab. Dispos., 2019, 47, (1), 1240.
15) D. S. On, P. Chertchinnapa, Y. Shinkai, T. Kojima and H. Nakano, "Development of a dual monoclonal antibody sandwich enzyme-linked immunosorbent assay for the detection of swine influenza virus using rabbit monoclonal antibody by Ecobody technology.", J. Biosci. Bioeng., 2020, DOI:10.1016/j.jbiosc.2020.03.003.
Q & A
-
Q
Can I use this kit for other proteins?
-
A
Yes, if the molecular weight is greater than 50,000.
-
Q
Do I have to use a Filtration tube prior to labeling the protein?
-
A
There is no need to use the Filtration tube if the following conditions are met.
a) The protein has disulfide groups and its solution does not contain small molecules with SH groups or disulfide groups. The concentration of the protein is 10 mg per ml or about 70 μM:Mix 10 μl protein solution and 100 μl Reducing agent solution, and follow the protocol starting at step 4.
b) The protein has SH groups and its solution does not contain small molecules with SH groups. The concentration of the protein is 10 mg per ml or about 70 μM: Mix 10 μl protein solution and 90 μl Reaction buffer, and add 8 μl SH-reactive biotin solution. Then follow the protocol starting at step 7.
-
Q
Do I have to use WS buffer to store the biotin-labeled protein?
-
A
You don’t have to use WS buffer. You can choose any kind of buffer according to your experiment.
-
Q
My sample contains small, insoluble material. What should I do?
-
A
Spin the sample and use the supernatant for the labeling.
-
Q
How long is the biotin-labeled protein stable?
-
A
If you store the biotin-labeled protein at 4ºC, it is stable for 2 months. For longer storage, add 100% volume of glycerol, aliquot, and store at -20ºC (if the protein can be frozen). However, please note that stability depends on the protein itself.
-
Q
What is the minimum amount of IgG that can be labeled by this kit?
-
A
The minimum amount is 10 μg. There is no significant difference in sensitivity and background between 10 and 100 μg of IgG.
Handling and storage condition
| 0-5°C, Protect from moisture | |
|
Danger / harmful symbol mark |

|
|---|---|