Detergent Screening Set (first choice-II)
Detergent
-
Product codeDS06 Detergent Screening Set (first choice-II)
| Unit size | Price | Item Code |
|---|---|---|
| 1 set | $202.00 | DS06-10 |
| 1 set | CHAPS, n-Dodecyl-β-D-maltoside, n-Octyl-β-D-glucoside, Sodium cholate(purifi |
200 mg x 1 200 mg x 1 200 mg x 1 200 mg x 1 200 mg x 1 |
|---|
Product Description
Detergent Screening Sets are used to find the best detergent for an application, such as enzyme stabilization, specific membrane protein isolation, assay development, protein crystallization, protein refolding, and so on. Detergent Screening Set (first choice-II) contains commonly used detergents for membrane protein isolation. Detergent Screening Set (for crystallization) contains detergents commonly used for protein crystallization.
Introduction
The phospholipid bilayer is the basic structure of the cell membrane. The most important functions of cells include transportation of substances, energy exchange, and transmission of information. These functions are conducted at the cell membrane by membrane proteins. In membrane biochemistry research, membrane proteins are solubilized and purified to study their structure and function. Proteins bound to cell membranes have hydrophobic sites buried within the phospholipid bilayers and hydrophilic sites facing toward the water layer. Detergents are used to isolate large insoluble molecules such as proteins. Detergents interact with the hydrophobic sites of proteins, which are then solubilized in the water layer, thus separating membrane proteins. It is important to choose a detergent that does not disrupt the bioactivities of target proteins. A detergent requires the following characteristics to be suitable for isolation of
membrane proteins:
1. Sufficient protein solubilization capability
2. No denaturing or inactivation of proteins
3. No interference with protein activities
4. No precipitation at 4ºC
5. Appropriate critical micelle concentrations (CMC) and micelle size
6. No absorption in the UV region
7. No toxicity
8. Availability of detergent detection methods
9. Non-ionic detergent if ion exchange chromatography is used
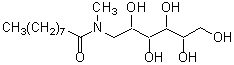
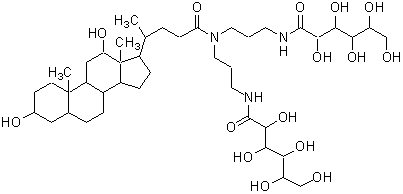
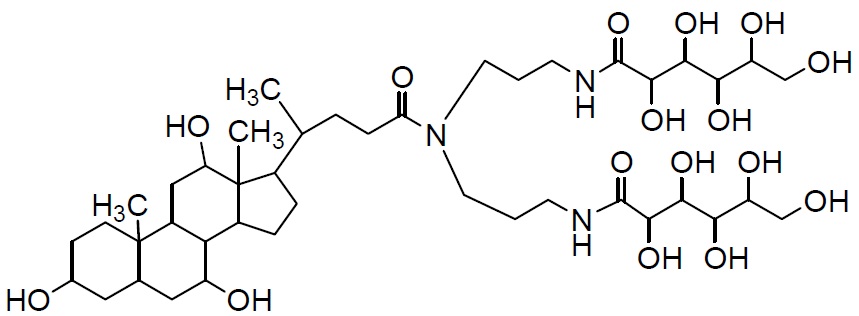
In the past, polyoxyethylene ether non-ionic detergents were widely used. These detergents, however, had several problems, such as denaturation of proteins and low CMC value, which cannot be separated easily by dialysis. n-Octyl-β-D-glucoside, n-Octyl-β-Dthioglucoside, CHAPS, and CHAPSO eliminate these problems and are widely used today. Most of the current detergents are non-ionic and easily applied to ion exchange chromatography purification. deoxy-BIGCHAP is a non-ionic detergent possessing deoxycholic acid and a gluconamide polar group. It has a high CMC value of 1.4 mM and can be easily separated by dialysis. Because its UV absorbance is low, it can be used for the determination of proteins. deoxy-BIGCHAP has been used for the extraction of opioid receptors from neuroblastoma or hybrid cells of glyoma. It has also been applied to adenylate cyclase or acetyltransferase. These detergents are also widely used to solubilize chromophores or to stabilize enzymes in diagnostic analyses and biochemical assays.
Trials of various kinds of detergents are needed to find the appropriate detergent for each study. Dojindo’s Detergent Screening Sets, which contain assorted packages of detergents, are available for use in these trials.
Technical info
Detergents are amphipathic compounds, with both lipophobic and lipophilic sites, that will form micelles above a critical concentration that is specific to each detergent. This is called the critical micelle concentration (CMC). The solubilizing abilities of detergents increase dramatically above their CMC values. After extracting membrane proteins, detergents can be easily removed by dilution, and then dialysis.
Table 1 Molecular Weight and Critical Micelle Concentration of Detergents
Specification
n-Dodecyl-β-D-maltoside: To pass the test
n-Octyl-β-D-glucoside: To pass the test
Sodium cholate(purified): To pass the test
MEGA-8: To pass the test
References
1) G. H. Perdew, H. W. Schaup and D. P. Selivonchick, "The Use of a Zwitterionic Detergent in Two-dimensional Gel Electrophoresis of Trout Liver Microsomes", Anal. Biochem, 1983, 135, 453.
2) Y. Matuo, N. Nishi, Y. Muguruma, Y. Yoshitaka, Y. Masuda, K. Nishikawa and F. Wada, "The Usefulness of CHAPS as a Non-cytotoxic Stabilizing Agent in Purification of Growth Factors", Cytotechnology, 1998, 1, 309.
3) R. Horiuchi, K Yamauchi, H. hayashi, S. Koya, Y. Takeuchi, K. Kato, M. Kobayashi and H. Takikawa, "Purification and Characterization of 55-kDa Protein with 3,5,3'-Triiodo-L-thyronine-binding Activity and Protein Disulfide-isomerase Activity from Beef Liver Membrane", Eur. J. Biochem., 1989, 183, 529.
4) W. F. Simods, G. Koski, R. A. Streaty, L. M. Hjelmeland and W. A. Klee, "Solubilization of Avtive Opiate Receptors", Proc. Natl. Acad Sci.USA, 1980, 77, 4623.
5) M. T. Lehto and F. J. Sharom, "Release of the Glycosylphoaphatidylinositol-anchoroesd Enzyme Ecto-5'-Nucleotidase by Phopholipase C: Catalytic Activation and Modulation by the Lipid Bilayer", Biochem. J., 1998, 332, 101.
6) K. Andersson, M. Hamalainen and M. Malmqvist, "Identification and Optimaization of Regeneration Conditions for Affinity-based Biosensor Assays. A Multivariate Cocktail Approach", Anal. Chem., 1999, 71, 2475.
7) R. Scindler, R. Mentlein and W. Feldheim, "Purification and Characterization of Retinyl Ester Hydrolase as a Member of the Non-specific Carboxylesterase Supergene Family", Eur. J. Biochem., 1998, 251, 863.
8) J. Cladera, J. L. Rigaud, J. Villaverde and M. Dunach, "Liposome Solubilization and Membrane Protein Reconstitution Using Chaps and Chapso", Eur. J. Biochem., 1997, 243, 798.
9) D. S. Liscia, T. Alhadi and B. K. Vonderhaae, "Solubilization of Active Prolactin Receptors by a Nondenaturing Zwitterionic Detergent", J. Biol. Chem., 1982, 257, 9401.
10) C. Yin, M. L. A. Ramos, M. I. B. Walmsley, R. W. Taylor, A. R. Walmsley, S. B. levu and P. A. Bullough, "The Quarternary Molecular Architecture of TetA, A Secondary Tetracycline Transporter from Escherichia Coli", Molecular Microbiology, 2000, 38, 482 .
11) R. Lutter, M. Saraste, H. S. Walraven, M. J. Runswick, M. Finel, J. F. Deatherage and J. E. Walker, "F1F0-ATP Synthase from Bovine Heart Mitochndria: Development of the Purification of a Monodisperse Oligomycin-sensitivr ATPase", Biochem. J., 1993, 295, 799.
12) J. Hendriks, A. Warne, U. Gohlke, T. Haltia, C. Ludovici, M. Lubben and M. Saraste, "The Active Site of the Bacterial Nitric Cxide Reductase Is a Dinuclear Iron center", Biochemistry, 1998, 37, 13102.
13) K. A. Williams, U. Geldmacher-Kaufer, E. Padan, S. Schuldiner and W. Kuhlbrandt, "Projection Structure of NhaA, a Secondary Transporter from Escherichia Coli, at 4.0Å Resolution", EMBO J., 1999, 18, 3558.
14) R. L. Krauth-Siegel, C. Sticherling, I. Jost, C. T. Walsh, E. F. Pai, W. Kabsch and B. Lantwin, "Crystallization and Preliminary Crystallographic Analysis of Trypanothione Reductase from Trypanosoma Cruzi, the Causative Agent of Chagas' Disease", FEBS Lett., 1993, 317, 105.
15) J. P. Allen, "Crystallization of the Reaction Center from Rhodobacter Sphaeroides in a New Tetragonal form", Proteins, 1994, 20, 283.
16) A. Shibata, N. Nakagawa, M. Sugahara, R. Masui, R. Kato, S. Kuramitsu and K. Fukuyama, "Crystallization and Preliminary X-ray Diffraction Studies of a DNA Excision Repair Enzyme, UvrB, from Thermus thermophilus HB8", Acta Cryst., 1999, D55, 704.
17) S. X. Lin, D. W. Zhu, A. Azzi, R. L. Campbell, R. Breton, F. Labrie, D. Ghosh, V. Pletnev, W. L. Duax and W. Pangborn, "Studies on the Three-dimensional Structure of Estrogenic 17 beta-hydroxysteroid Dehydrogenase", J. Endocrinol., 1996, 150, S13.
18) H. Tokuda, J. Akimaru, S. Matsuyame, K. Nishiyama and S. Mizushima, "Purification of SecE and Reconctitution of SecE-dependent Protein Translocation Activity", Fed. Eur. Biochem. Soc., 1991, 279, 233.
19) 土屋友房,“膜タンパク質の可溶化と界面活性剤”, 化学と生物実験ライン5, 廣川書店, 1990.
20) M. J. Newman, D. L. Foster, T. H. Wilson and H. R. Kaback, "Purification and Reconstitution of Functional Lactose Carrier From Escherichia Coli", J. Biol. Chem., 1981, 256, 11804.
21) M. Nishikawa, F. Komada, Y. Uemura, H. Hidaka and S. Shirakawa, "Decreased Expression of Type II Protein Kinase C in HL-60 Variant Cells Resistant to Cell Differentiation by Phorbol Diester", Cancer Res., 1990, 50, 621.
22) J. R. Wright, D. F. Zlogar, J. C. Taylor, T. M. Zlogar and C. I. Restrepo, "Effects of Endotoxin on Surfactant Protein A and D Stimulation of NO Production by Alveolar Macrophages", Am. J. Physiol., 1999, 276, 650.
Handling and storage condition
| 0-5°C, Protect from moisture |