General Information
Ab-10 Rapid Biotin Labeling Kit enables rapid (in less than 30 min) and easy labeling of Biotin to 10 μg antibody.Reactive Biotin (a component of the kit) has a succinimidyl ester group, that can easily make a covalent bond with an amino group of the target antibody without any activation process. This kit contains all the necessary reagents to prepare a Biotin-labeled antibody except for DMSO.
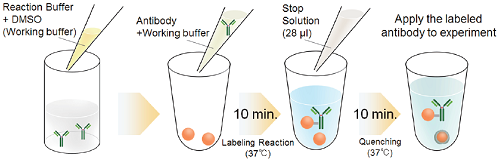
Fig. 1 Labeling procedure
|
Caution After a Reactive Biotin is taken out from the seal bag, keep the unused Reactive Biotin in the bag, seal tightly and store at -20°C. Store the other components at 0-5°C. |
Kit Contents
| Reactive Biotin* | ×3 |
| Reaction Buffer | 100 μl ×1 |
| Stop Solution | 100 μl ×1 |
- Though it is hard to see the reagent Reactive Biotin because of its small amount, it is attached on the bottom of the tube as a sheet form. Please collect the Reactive Biotin carefully by pipetting with buffer as described in Step 4 of the protocol.
Storage Condition
Store at 0-5 °C
This kit is stable for 1 year at 0-5°C before opening.
Required Equipment and Materials
- 20 μl, 200 μl adjustable pipettes
- Microtube (for sample and Working buffer preparation)
- Incubator (37 °C)
- PBS (Phosphate buffered saline)
- DMSO (Dimethyl sulfoxide)
Precaution
- Use 0.5-1 mg/ml of antibody solution for labeling. If the antibody concentration is more than 1 mg/ml, please dilute the antibody solution with PBS.
- If the sample solution contains small insoluble materials, centrifuge the solution, and use the supernatant for the labeling.
- The microtubes in this kit contain solutions. Since there is a possibility that the droplets might attach to the inside walls or caps, please spin the tube to drop them down prior to open.
- In case an antibody solution includes a high concentration of constituents, such as BSA or glycerol, it may interfere with a labeling and cause a non-specific signal. We recommend removing the constituents prior to labeling procedure. Usable constituents (○) and non-usable constituents (×) are shown in Table 1, and compatible concentrations of constituents are shown in Table 2.
Table 1. Usable/non-usable constituents
| Additivea | |
| Buffering agents (PBS,HEPES) | 〇 |
| Sodium chloride | 〇 |
| Chelating agents (EDTA) | 〇 |
| Sodium azide | 〇 |
| Sugars (Glucose, Trehalose) | 〇 |
| Primary amines and thiols | × |
| Glycerol | BSA | Gelatin | Tris | |
| Anti-Chloramphenicol Acetyl Transferase (CAT) antibody | < 20% | < 0.1% | < 0.1% | < 50 mmol/l |
| Anti-GAPDH antibody | < 50% | < 0.1% | < 0.1% | < 50 mmol/l |
| Anti-CD44 antibody | < 50% | < 0.5% | < 0.1% | < 50 mmol/l |
- Interference and non-specific signal may be dependent on types of antigen, host species of antibody or constituents.
Protocol
- Add Reaction Buffer (up to 30 μl) to a microtube and mix it with an equal volume of DMSO to prepare Working buffer.
- Add 0.5-1 mg/ml of the antibody solution to another microtube to be an amount of antibody of 10 μg.
- Add Working buffer (step.1) to the antibody solution (step 2) and mix by pipetting.
- The volume of Working buffer : one-fifth of the antibody solution (Table 3).
- Add the solution (step 3) to Reactive Biotin and mix by pipetting.
- Incubate at 37°C for 10 minutes.
- Add 28 μl of Stop Solution to the solution (step 5) and mix by pipetting.
- Incubate at 37°C for 10 minutes.
- Apply the sample (step 7) for desired experiments or store at 0-5°C.
- The labeled antibody is stable at 4oC for 2 weeks.
| The concentration of antibody (mg/ml) |
0.5 | 0.6 | 0.7 | 0.8 | 0.9 | 1.0 |
| The volume of Working buffer (μl) |
4.00 | 3.34 | 2.86 | 2.50 | 2.22 | 2.00 |
Supplimental Information
Mitochondria immunostaining
- HeLa cells were seeded on a μ-slide 8 well (ibidi) and cultured overnight at 37 °C in a 5% CO2 incubator.
- The cells were washed with PBS three times, and 4% paraformaldehyde in PBS was added to the μ-slide.
- The μ-slide was incubated at room temperature for 15 minutes.
- The cells were washed with PBS three times, and 1% Triton-X in PBS was added to the μ-slide.
- The μ-slide was incubated at room temperature for 30 minutes.
- Once the cells were washed with PBS three times, a blocking solution prepared with PBS was added to the μ-slide.
- The cells were then incubated at room temperature for 1 hour.
- Biotin conjugated anti-mitochondria antibody was diluted 50 times with the blocking solution.
- Anti-mitochondria antibody was purchased from Abcam (Product Code: ab3298) .
- The supernatant was discarded and the solution (step 8) was added to the μ-slide.
- The μ-slide was incubated at 0-5oC overnight.
- The supernatant was discarded and the cells were washed using PBS-T three times.
- 0.2 μg/ml peroxidase conjugated streptavidin was added to the μ-slide.
- The μ-slide was incubated at room temperature for 1 hour.
- The supernatant was discarded and the cells were washed using PBS-T three times.
- The cells were washed using Tris buffer (TB, 50 mmol/l, pH 7.5) three times.
- The supernatant was discarded and DAB solution [0.2 mg/ml DAB (Dojindo Laboratories, Product Code:D006),
0.003% H2O2 , 50 mmol/l Tris (pH 7.5)] was added to the μ-slide. - The μ-slide was incubated at room temperature for 10 minutes.
- After the cells were washed using TB three times, TB was added to the μ-slide.
- The cells were observed under a microscope.
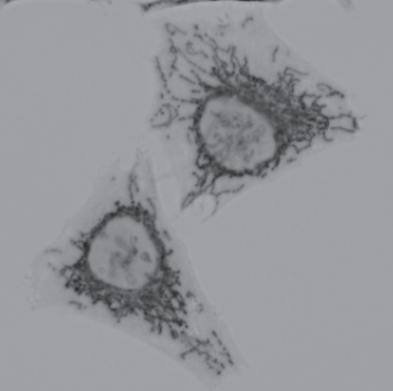
Fig. 2 Microscope image of DAB-stained mitochondria in HeLa cells
Frequently Asked Questions / Reference
LK37: Ab-10 Rapid Biotin Labeling Kit
Revised Nov., 29, 2023


 Hidden sections will not be printed.
Hidden sections will not be printed.

