| p53 activation contributes to tumor suppression in cancer and can induce cellular senescence depending on context. This article highlights recent findings on p53-driven senescence and its relationship to lysosome and autophagy pathways. Recent studies showed that, in senescent cells, p53 promotes autophagy- and lysosome-dependent lipid recycling, increasing phosphoethanolamine supply to meet phospholipid demand from cellular hypertrophy and organelle remodeling. In a lung cancer model, p53 reactivation induced a TSC2-mTORC1-TFEB cascade that upregulated lysosome and autophagy programs, leading to autophagy-dependent senescence and tumor regression through macrophage-mediated clearance of senescent cells. Together, these findings advance our understanding of p53-driven senescence and indicate that lysosome and autophagy pathways contribute to this process. | ||||||||||||||||||
|
p53 increases phospholipid headgroup scavenging in senescence (Nature Cell Biology, 2026) Highlighted technique: SA-βGal , a senescence marker based on lysosome-derived β-galactosidase activity, was used to confirm and quantify senescence under p53 ON/OFF and PE synthesis–inhibiting conditions. p53 was controlled by Nutlin-3a–mediated activation or dox-based p53 ON/OFF regulation, and the senescent fraction was quantified by counting the percentage of SA-βGal–positive cells. |
||||||||||||||||||
|
p53 Drives Lung Cancer Regression through a TSC2/TFEB-dependent Senescence Program (Cancer Discovery, 2026) Highlighted technique: The authors tested whether p53 reactivation truly increases autophagy activity in lung cancer cells by monitoring LC3-II, an autophagosome-associated marker, and the lysosomal marker LAMP1. After confirming that p53 elevated LC3-II and LAMP1, they added bafilomycin and observed further LC3-II accumulation, supporting active autophagic flux rather than impaired degradation under p53 reactivation. |
||||||||||||||||||
Senescence, Autophagy and Lysosome Indicators (click to open/close)
|
||||||||||||||||||
Application Note (click to open/close)
|
||||||||||||||||||
|
|
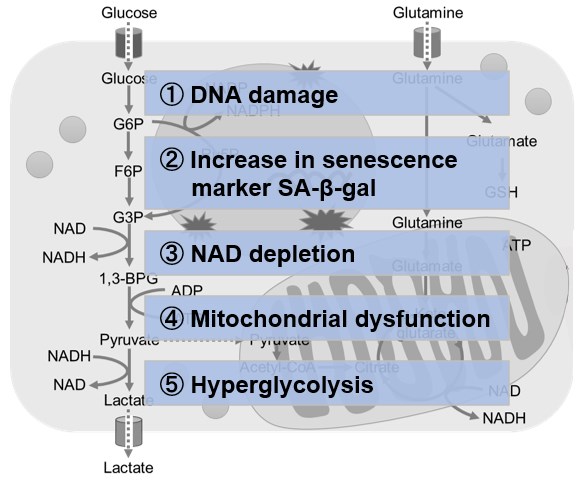
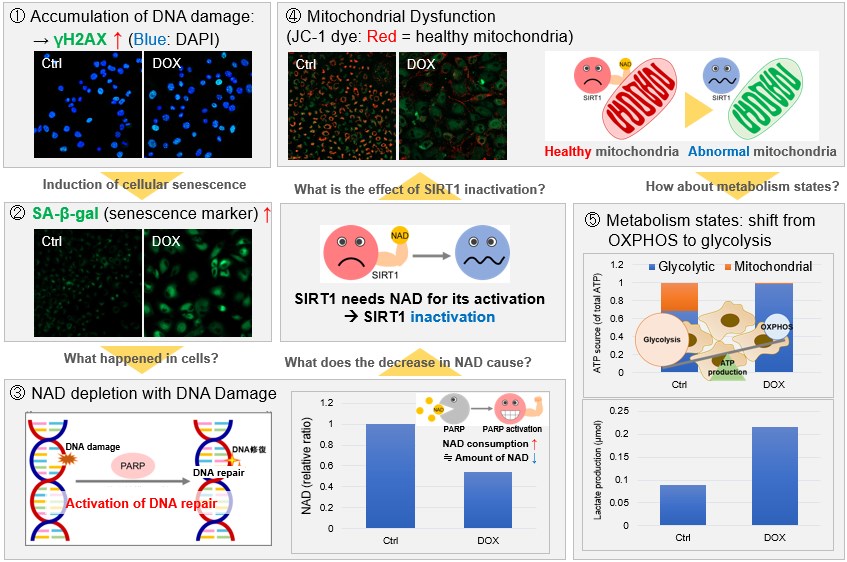
NAD(+) levels decline during the aging process, causing defects in nuclear and mitochondrial functions and resulting in many age-associated pathologies*. Here, we try to redemonstrate this phenomenon in the doxorubicin (DOX)-induced cellular senescence model with a comprehensive analysis of our products. *S. Imai, et al., Trends Cell Biol, 2014, 24, 464-471
|
 |
|