|
Altered cellular metabolism within the tumor microenvironment is a primary characteristic supporting cancer growth. Investigating the cross-talk between immune cells and metabolic pathways is important for understanding tumor progression. One recent study showed that the inflammasome adapter protein ASC links innate immunity and metabolic reprogramming to drive pancreatic adenocarcinoma. Another study demonstrated that tumor-associated microglia upregulate oxidative phosphorylation to secrete extracellular ATP, fueling glioblastoma proliferation. These findings indicate that specific molecular mediators linking innate immune responses to mitochondrial energy production directly facilitate cancer progression.
|
|
Cancer cell-intrinsic inflammasome protein ASC links innate immunity with mitochondrial metabolism in driving pancreatic cancer (Nature Communications, 2026)
Summary: This study shows that the inflammasome adapter protein ASC acts as a molecular bridge linking innate immunity, mitochondrial dysfunction, and metabolic reprogramming to drive pancreatic ductal adenocarcinoma (PDAC) progression. Specifically, upregulated ASC and Caspase-1 in the pancreatic epithelium promote tumorigenesis. Mechanistically, global or epithelial-specific ASC deletion, or targeting extracellular ASC, suppresses pancreatic tumor formation.
Highlighted technique: To evaluate ASC-driven metabolic reprogramming, the authors measured cellular Oxygen Consumption Rate (OCR) utilizing the Seahorse XF analyzer. Combined with whole transcriptome profiling, they explicitly revealed how ASC alters mitochondrial respiration to fuel tumor progression.
Dojindo's OCR assay kit enables plate reader-based measurements using fewer cells. It shows lower running costs, and can be applied for preliminary tests before Seahorse measurement.
|
|
Tumor-Associated Microglia Secrete Extracellular ATP to Support Glioblastoma Progression (Cancer Research, 2024)
Summary: This study shows that glioblastomas establish extracellular ATP (eATP) that directly fuels tumor progression. Mechanistically, glioma-secreted GM-CSF induces mitochondrial fission in TAMs, leading to augmented eATP release that promotes cancer cell proliferation via the P2X7 receptor.
Highlighted technique: To evaluate microglia-derived metabolic support, the authors measured secreted ATP and proliferation in co-cultured glioma cells and microglia treated with P2X7 antagonists to block eATP signaling and demonstrate that glioma proliferation is directly dependent on the activation of the P2X7 receptor.
Here, it is an extracellular ATP assay kit designed to evaluate unstable ATP using simple procedures and a detailed protocol.
|
Metabolism Activity Assay (click to open/close)
|
Application note (click to open/close)
Comparison of metabolic pathway in two types of cancer cells
The dependence of OXPHOS and Glycolysis in two types of cancer cells, HeLa and HepG2, were compared based on Lactate production, ATP levels, and OCR values.
Many cancer cells produce ATP through the glycolytic pathway. On the other hand, it has been recently reported that cancer cells whose glycolytic pathway is suppressed survive by shifting their energy metabolism to OXPHOS by enhancing mitochondrial function, and the dependency of metabolic pathways differs depending on cell lines.
|
<Evaluation by Lactate production and ATP levels>
-
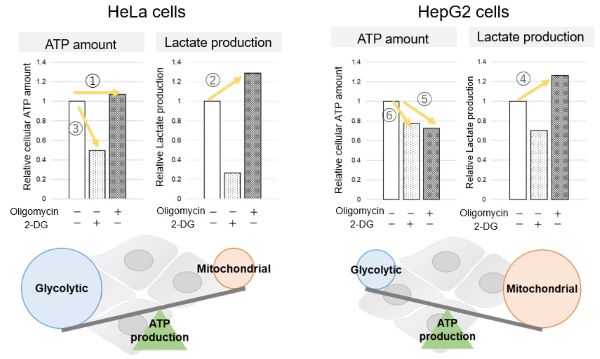
We confirmed the changes in ATP and Lactate production when ATP synthesis by OXPHOS was inhibited by Oligomycin stimulation and by 2-Deoxy-D-glucose (2-DG) in the glycolytic pathway. The results showed that HeLa cells depend on Glycolysis and HepG2 cells depend on OXPHOS to synthesize ATP.
-
When OXPHOS was inhibited in HeLa cells, ATP levels remained unchanged (①), and lactate production increased (②). This suggests that even when OXPHOS is inhibited, glycolysis can be further activated. Conversely, when glycolysis is inhibited, ATP levels decrease significantly (③), indicating that energy production depends on glycolysis. On the other hand, when OXPHOS was inhibited in HepG2 cells, lactate production increased (④), indicating that the cells attempt to compensate for energy production by enhancing glycolysis, but ATP levels still decrease (⑤). This means that even with increased glycolysis, ATP production is not sufficiently compensated. Furthermore, ATP levels decrease more when glycolysis is inhibited (⑥), suggesting that energy production in HepG2 cells depends more on OXPHOS than glycolysis.
Products in Use
- Glycolysis/OXPHOS Assay Kit
-
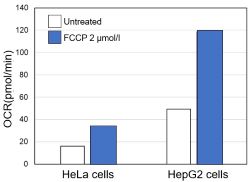
<Evaluation by OCR value>
-
Using the same number of cells, we measured the OCR value when cellular oxygen consumption was promoted by stimulating the cells with FCCP, a mitochondrial uncoupling agent. The results showed that HepG2 cells had higher OCR values than HeLa cells, suggesting a greater dependence on OXPHOS, correlating with the results obtained from ATP level and Lactate production.
Products in Use
- Extracellular OCR Plate Assay Kit
|
|


















