|
Lactate, best known as a metabolite in the body, plays several key roles. It is primarily produced during anaerobic metabolism, such as the Warburg effect in cancer cells, as a byproduct of the conversion of glucose to energy when oxygen levels are low. Beyond its role in energy metabolism, lactate acts as a signaling molecule, influencing various cellular processes such as gene expression and inflammation regulation. In addition, recent research suggests that lactate may serve as an alternative energy source for the brain and other tissues, challenging the traditional view of lactate as a waste product.
|
-
Metabolic regulation of homologous recombination repair by MRE11 lactylation
Click here for the original article: Yuping Chen, et. al., Cell, 2024.
Point of Interest
- MRE11 undergoes lactylation by CBP in response to DNA damage, a process that is dependent on ATM.
- The lactylation of MRE11 facilitates DNA end breakage and homologous recombination repair.
- Elevated lactate levels in cancer cells lead to the lactylation of MRE11, contributing to chemoresistance.
- Targeting the lactylation of MRE11 can increase the sensitivity of cancer cells to chemotherapy.
-
Lactate modulates iron metabolism by binding soluble adenylyl cyclase
Click here for the original article: Wei Liu, et. al., Cell Metabolism, 2023.
Point of Interest
- Lactate promotes the expression of hepatic hepcidin via the cAMP-PKA-Smad signaling pathway.
- Lactate directly binds to soluble adenylyl cyclase, enhancing its enzymatic activity.
- Lactate modulates iron homeostasis by inducing the expression of hepatic hepcidin.
-
Lactate activates the mitochondrial electron transport chain independently of its metabolism
Click here for the original article: Xin Cai, et. al., Molecular Cell, 2023.
Point of Interest
- Lactate acts as a mitochondrial messenger, promoting the shift of ATP production to the mitochondria.
- It activates the electron transport chain (ETC) independently of its own metabolism.
- Activation of the ETC by lactate results in increased pyruvate oxidation and enhanced lactate utilization.
- The mitochondrial ETC can sense the availability of lactate and recognize it as a potential nutrient.
|
| Related Techniques |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Related Applications |
Comparison of metabolic pathway in two types of cancer cells
The dependence of OXPHOS and Glycolysis in two types of cancer cells, HeLa and HepG2, were compared based on Lactate production, ATP levels, and OCR values.
Many cancer cells produce ATP through the glycolytic pathway. On the other hand, it has been recently reported that cancer cells whose glycolytic pathway is suppressed survive by shifting their energy metabolism to OXPHOS by enhancing mitochondrial function, and the dependency of metabolic pathways differs depending on cell lines.
|
<Evaluation by Lactate production and ATP levels>
-
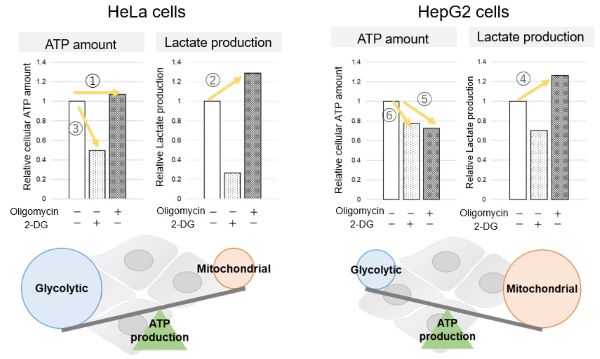
We confirmed the changes in ATP and Lactate production when ATP synthesis by OXPHOS was inhibited by Oligomycin stimulation and by 2-Deoxy-D-glucose (2-DG) in the glycolytic pathway. The results showed that HeLa cells depend on Glycolysis and HepG2 cells depend on OXPHOS to synthesize ATP.
-
When OXPHOS was inhibited in HeLa cells, ATP levels remained unchanged (①), and lactate production increased (②). This suggests that even when OXPHOS is inhibited, glycolysis can be further activated. Conversely, when glycolysis is inhibited, ATP levels decrease significantly (③), indicating that energy production depends on glycolysis. On the other hand, when OXPHOS was inhibited in HepG2 cells, lactate production increased (④), indicating that the cells attempt to compensate for energy production by enhancing glycolysis, but ATP levels still decrease (⑤). This means that even with increased glycolysis, ATP production is not sufficiently compensated. Furthermore, ATP levels decrease more when glycolysis is inhibited (⑥), suggesting that energy production in HepG2 cells depends more on OXPHOS than glycolysis.
Products in Use
- Glycolysis/OXPHOS Assay Kit
-
<Evaluation by OCR value>
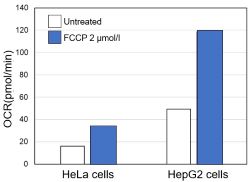 
-
Using the same number of cells, we measured the OCR value when cellular oxygen consumption was promoted by stimulating the cells with FCCP, a mitochondrial uncoupling agent. The results showed that HepG2 cells had higher OCR values than HeLa cells, suggesting a greater dependence on OXPHOS, correlating with the results obtained from ATP level and Lactate production.
Products in Use
- Extracellular OCR Plate Assay Kit
|


















