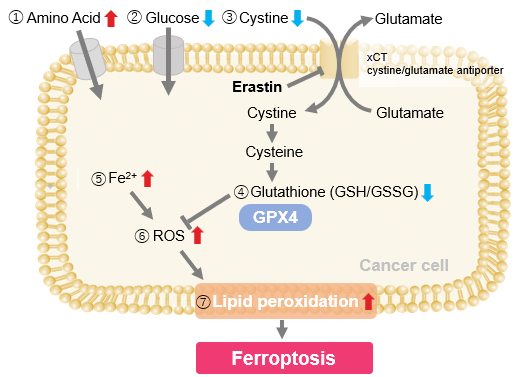
| Ferroptosis is considered a promising therapeutic strategy for cancer, and control of the lipid peroxidation that drives this process is therefore of major importance. Cancer cells influence ferroptosis sensitivity by regulating lipid availability and iron metabolism. Clarifying how tumors control these processes is important for understanding treatment resistance and for identifying metabolically targetable vulnerabilities. Recent studies showed that metabolites derived from peritumoural adipose tissue suppress ferroptosis by inhibiting ferritinophagy in cancer cells. Another study showed that acidic tumors remodel the glycocalyx into a chondroitin sulfate rich surface barrier, thereby limiting extracellular lipid uptake while promoting lipid droplet formation to reduce ferroptotic stress. Together, these findings highlight microenvironment dependent regulation of lipid and iron metabolism as an important determinant of ferroptosis responses in tumors. | ||||||||||||||||||||||||||
|
1. Peritumoural adipose tissue promotes ferroptosis resistance by 3-hydroxykynurenine-mediated suppression of ferritinophagy (Nature Cell Biology, 2026) Highlighted technique: To test whether PAT and kynurenine-pathway metabolites alter ferroptosis-related oxidative damage and iron availability, the authors quantified lipid peroxidation in treated cancer cells using C11-BODIPY staining followed by flow cytometry. They also assessed intracellular ferrous iron using an iron assay kit, as well as FerroOrange-based detection by imaging or flow cytometry. |
||||||||||||||||||||||||||
|
2. Tumour acidosis remodels the glycocalyx to control lipid scavenging and ferroptosis(Nature Cell Biology, 2026) Highlighted technique: To examine lipid accumulation in acidic tumors, the authors visualized lipid droplets in patient tumor sections, patient-derived cells, and 3D spheroids using fluorescent probe–based imaging. They then fluorescently labeled extracellular vesicles, LDL, and HDL, and measured their binding to or uptake by cells with confocal microscopy and flow cytometry to assess extracellular lipid particle uptake. |
||||||||||||||||||||||||||
Ferroptosis Indicators (click to open/close)
|
||||||||||||||||||||||||||
Application Note (click to open/close)
|
||||||||||||||||||||||||||
|
|
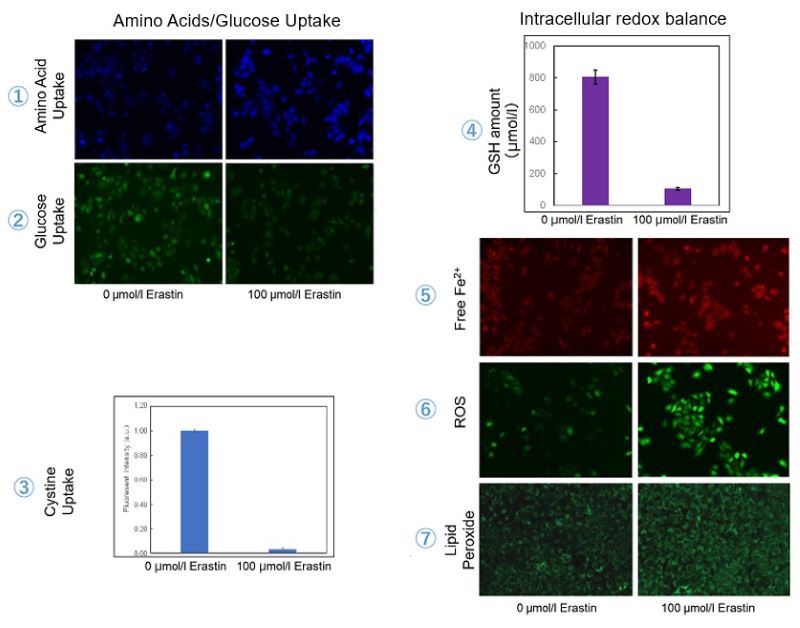
We investigated the transition of cellular metabolisms in A549 cells treated with erastin, a known ferroptosis inducer. Our results revealed the following. Results Cell Line: A549 |
||
|
Products in Use |
 |
||