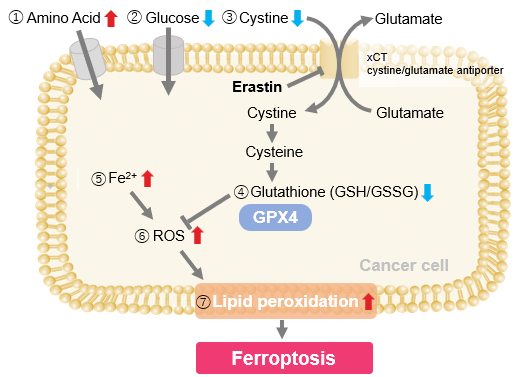
| Ferroptosis is a form of cell death driven by lipid peroxidation, and its execution may be influenced by lysosomal function. Clarifying how lysosomes regulate ferroptosis sensitivity and cell death is important for understanding tumor responses and the control of cell death. Recent studies showed that, in non-small cell lung cancer, SMARCA4 deficiency confers ferroptosis resistance through a SMARCA4-ALDH16A1-TXN pathway involving lysosome mediated thioredoxin degradation. Another study showed that GPX4 inhibition gives rise to both apoptotic-like and necrotic-like cells during ferroptosis, and that lysosomal rupture, glutathione depletion, and cathepsin activity promote necrotic-like outcomes. These findings deepen our understanding of the relationship between lysosomes and ferroptosis. | ||||||||||||||||||||||||||||
|
1. Targeting ALDH16A1 mediated thioredoxin lysosomal degradation to enhance ferroptosis susceptibility in SMARCA4-deficient NSCLC (Nature Communications, 2025) Highlighted technique: To determine whether ALDH16A1 promotes lysosomal degradation of thioredoxin (TXN), the authors first examined whether TXN is regulated at the RNA or protein level in cells with altered ALDH16A1 expression. They then blocked new protein synthesis and added chloroquine or MG132 to test whether TXN is degraded through the lysosomal or proteasomal pathway. Finally, they visualized TXN and the lysosomal marker LAMP1 by immunofluorescence to assess whether ALDH16A1 loss or restoration changes TXN–lysosome colocalization. |
||||||||||||||||||||||||||||
|
2. Ferroptosis induces heterogeneous death profiles that are controlled by lysosome rupture (Developmental Cell, 2026) Highlighted technique: To determine whether lysosomal rupture occurs during GPX4 inhibition-induced cell death, the authors used cells expressing the fluorescent reporter GFP-Gal3 and monitored punctate signals that appear upon lysosomal damage. By tracking GFP-Gal3 together with Sytox staining after GPX4 inhibition, they examined whether lysosomal rupture occurs before plasma membrane rupture and whether it differs between cells showing necrotic or apoptotic features. |
||||||||||||||||||||||||||||
Ferroptosis Indicators (click to open/close)
|
||||||||||||||||||||||||||||
Application Note (click to open/close)
|
||||||||||||||||||||||||||||
|
|
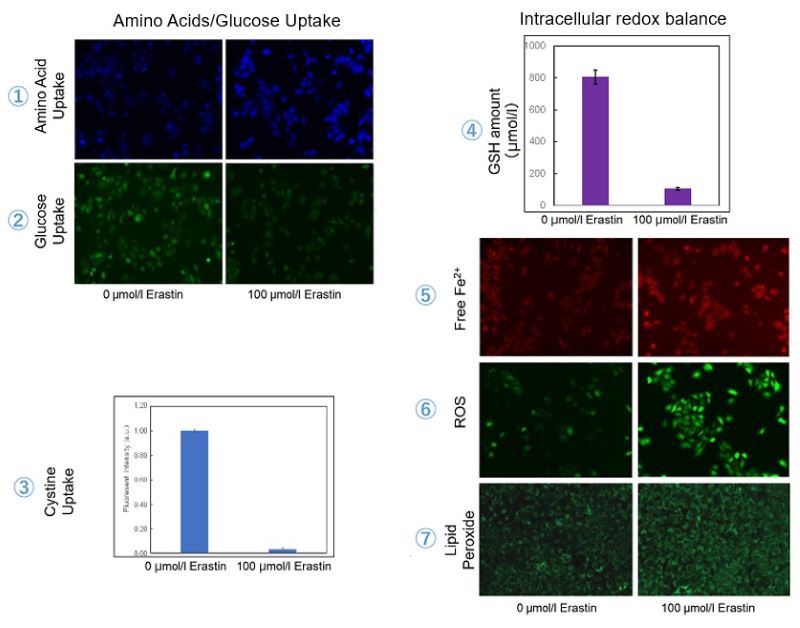
We investigated the transition of cellular metabolisms in A549 cells treated with erastin, a known ferroptosis inducer. Our results revealed the following. Results Cell Line: A549 |
||
|
Products in Use |
 |
||